Australian clinical guideline for diagnosing and managing acute coronary syndromes 2025

Acute management of STEMI – reperfusion for STEMI
Ongoing management of fibrinolytic-treated people
Acute management of NSTEACS
Antiplatelet therapy in the acute phase
Anticoagulant therapy in the acute phase
Acute management of ACS with cardiac arrest and/or cardiogenic shock
Treatment for ACS with multivessel disease without cardiogenic shock
Coronary artery bypass graft surgery in ACS
Treatment for SCAD
Myocardial infarction with non-obstructive coronary arteries
MI due to oxygen supply/demand mismatch without acute coronary occlusion
Echocardiography
Duration of cardiac monitoring
 2. Hospital care and reperfusion
2. Hospital care and reperfusion
Acute management of STEMI – reperfusion for STEMI
Eligibility for reperfusion
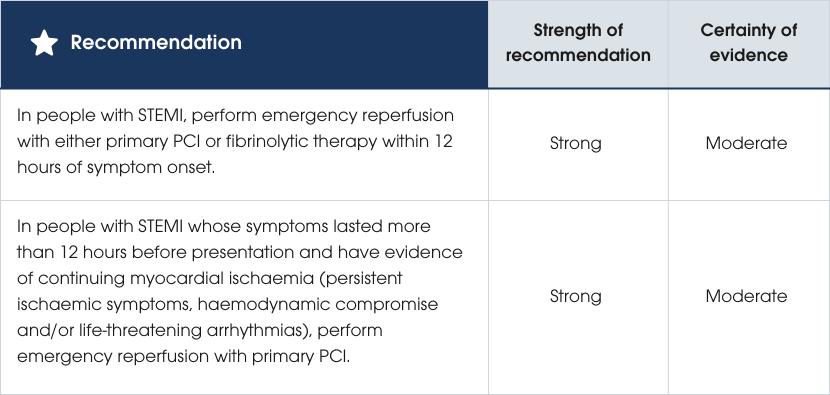
Recommendations
Recommendations
Evidence supporting the recommendations
Practice points
- Assess cognitive function, comorbidities and frailty when determining eligibility for reperfusion, as these factors significantly influence overall survival outcomes.
- Consider percutaneous coronary intervention (PCI) as it shows advantages over fibrinolysis in older populations. However, note that evidence is limited by small sample sizes, a lack of data for individuals over 90 years, and the absence of assessments for frailty or comorbidities in supporting trials.
Evidence supporting the recommendations
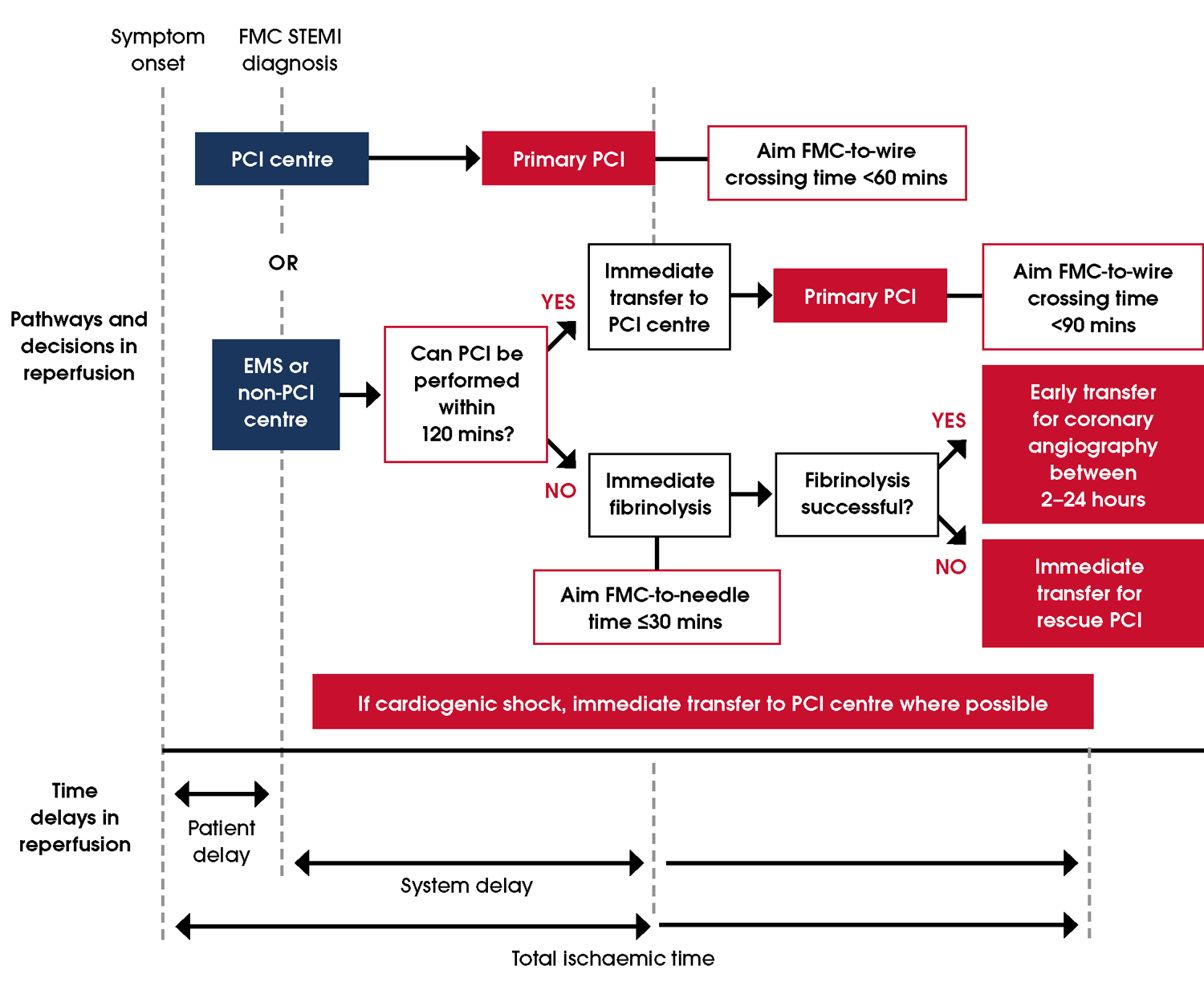
Reperfusion for STEMI involves either primary PCI or fibrinolytic therapy, with PCI preferred if it can be performed within 120 minutes of first medical contact. At PCI-capable centres, wire crossing should occur within 60 minutes, or within 90 minutes for people who have been transferred [256]. Fibrinolysis reduces 35-day mortality compared with no treatment [257]. However, PCI is more effective than fibrinolysis in reducing short- and long-term risks of death, non-fatal reinfarction and stroke. Early fibrinolysis followed by angiography may be comparable to PCI, particularly if initiated within two hours of symptom onset [248]. Administering fibrinolysis very early, including pre-hospital administration, may result in better outcomes than PCI for people presenting within two hours of symptom onset [236]. Fibrinolysis is not recommended after 12 hours post-symptom-onset; instead, PCI is preferred for people with ongoing myocardial ischaemia [254, 255, 257–263]. For a decision-making chart see Figure 12. Figure 12 Decision making and organisation of reperfusion strategies within first 12 hours of medical contact. Adapted from Chew et al. [22] Abbreviations: EMS, emergency medical service; FMC, first medical contact; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction. Evidence supporting the recommendations
Practice points
- Establish effective care pathways tailored to the specific healthcare services available in the region. These pathways should include ambulance, primary health, emergency, cardiology and regional or remote healthcare services to optimise reperfusion times.
- Implement specific measures to reduce time to reperfusion [264], such as:
- pre-hospital ECG and single-call catheter laboratory activation (i.e. streamlined communication between emergency services and the receiving hospital to bypass the ED and reduce reperfusion delay)
- pre-hospital fibrinolysis by suitably trained clinicians (e.g. paramedics, nurses, First Nations health practitioners)
- direct transfer to PCI-capable hospitals and direct transfer to the catheterisation laboratory on hospital arrival.
Evidence supporting the recommendations
Evidence supporting the recommendations
Timing of fibrinolytic therapy
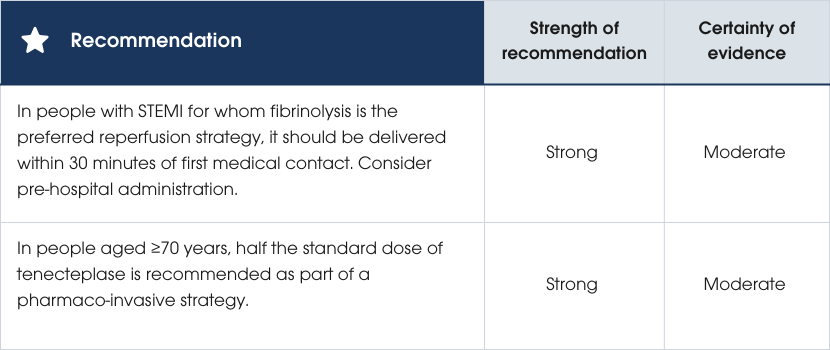
Fibrinolysis should be considered when primary PCI is delayed by more than 120 minutes and there are no absolute contraindications. It should be administered as soon as possible, ideally within 30 minutes of first medical contact, and, if feasible, before hospital arrival. People with absolute contraindications should be transferred for PCI (Table 9). People with a relative contraindication need to have the risks and benefits of treatment considered [256, 264–266].Dosing fibrinolytic therapy in older people
A comparison of pre-hospital fibrinolysis with angiography 6–24 hours later against primary PCI in people unable to receive the PCI within 60 minutes showed no significant difference in outcomes such as death, cardiogenic shock, heart failure or recurrent MI. In people aged 75 and older, full-dose tenecteplase was associated with higher rates of intracranial haemorrhage, a risk reduced by halving the dose without affecting efficacy [265]. Further research in older people (over 60 years, mean age 70 years) confirmed similar efficacy between half-dose tenecteplase and routine angiography 6–12 hours later against primary PCI, although intracranial haemorrhage was slightly higher in the fibrinolysis group (1.5% vs 0%), half of these events linked to dosing errors [267].Practice points
- An easily administrable fibrinolytic agent that can be given as a bolus dose, such as tenecteplase, is advisable, especially in the pre-hospital setting.
- Currently available fibrinolytics include:
- tenecteplase (weight-adjusted [30–50 mg] IV bolus and age-adjusted [half-dose] IV bolus)
- reteplase (10 units IV followed by 10 units IV, 30 minutes later)
- alteplase (weight-adjusted accelerated bolus and infusion regimen).
Procedural recommendations in primary percutaneous coronary intervention
Recommendations
Recommendations
Evidence supporting the recommendations
Evidence supporting the recommendations
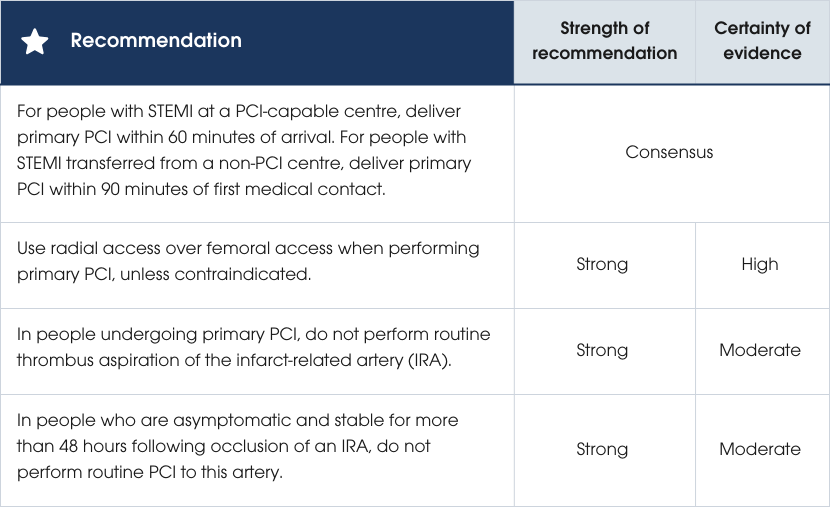
Time targets for primary PCI
For people diagnosed with STEMI, reducing treatment delays from first medical contact to reperfusion is crucial for improving mortality outcomes [268]. Recommended targets include primary PCI within 60 minutes for those arriving at a PCI-capable centre, or within 90 minutes for those transferred from a non-PCI-capable centre [256]. Pre-hospital diagnosis and direct activation of the catheterisation laboratory, and bypassing ED on arrival, can minimise delays [269, 270].Radial versus femoral access
Radial access is preferred for primary PCI in STEMI due to its association with lower mortality (1.6% vs 2.1%) and major bleeding (1.5% vs 2.7%) compared to femoral access [271–275]. A radial-first approach is recommended unless contraindicated [118, 265, 271, 276].Treatment of the infarct-related artery
Thrombus aspiration of the infarct-related artery (IRA) carries a small increased risk of stroke without survival benefits and may be considered for individuals with a high thrombus burden. Technical strategies to minimise embolisation should be employed [277–279]. When stenting is required, drug-eluting stents are preferred over bare metal stents due to lower rates of restenosis and stent thrombosis, including in individuals at high bleeding risk, those requiring triple antithrombotic therapy or short-duration dual antiplatelet therapy (DAPT) [256, 280–285]. Routine deferred stent implantation in people with STEMI does not improve outcomes compared with standard immediate stent implantation, and the need for unplanned target vessel revascularisation may be increased. However, it may be considered in cases of significant thrombus burden where immediate PCI is unlikely to succeed [286–289]. Routine PCI of a completely occluded IRA beyond 48 hours in asymptomatic, stable individuals is not advised, as it may increase the risk of recurrent MI without improving survival or major cardiovascular outcomes [260, 290].Practice points
- Where stenting is required, drug-eluting stents are preferred over bare metal stents.
- Routine deferred stenting of the IRA is not recommended. In people with STEMI and risk factors for slow or no reflow, such as high thrombus burden, consider deferred stent implantation.
- In people with STEMI where primary PCI of the IRA is not feasible (e.g. severe left main artery disease or an uncrossable coronary lesion), CABG may be an appropriate primary reperfusion strategy. CABG may be particularly appropriate if there is a large area of myocardium at risk and surgery is available in a timely manner (refer to Section Coronary artery bypass graft surgery in ACS) [291, 292].
Women
- Understand that women with STEMI have documented delays to reperfusion, lower rates of invasive angiography and radial access as well as poorer outcomes compared with men [14–16].
- Improve clinician awareness of sex-specific differences in:
- presenting symptoms
- ECG diagnostic criteria
- underlying MI aetiologies, such as a higher prevalence of SCAD and MINOCA, as these may improve outcomes.
- Perform primary PCI as the preferred revascularisation strategy in pregnant women with STEMI, except when caused by SCAD. Use appropriate shielding to minimise radiation exposure to the foetus [276].
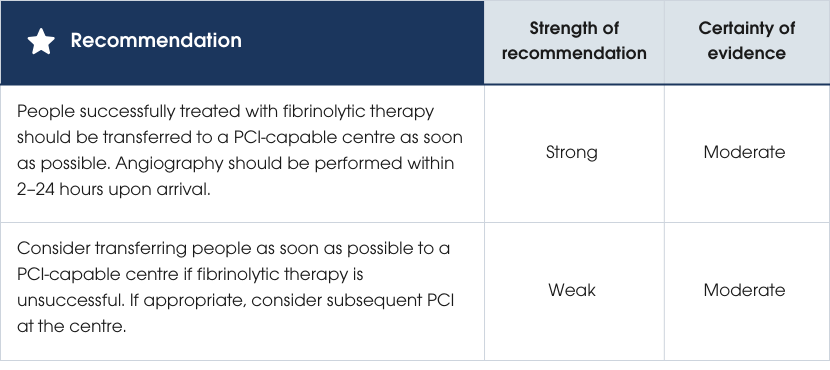
Ongoing management of fibrinolytic-treated people
Recommendations
Recommendations
Evidence supporting the recommendations
Primary PCI is associated with the lowest mortality compared to fibrinolysis alone [293]. Among individuals receiving initial fibrinolysis, a pharmaco-invasive approach (PCI ≥2 hours after fibrinolysis) reduces reinfarction and trends towards lower mortality compared to fibrinolysis alone or facilitated PCI (<2 hours) [293]. A Bayesian analysis further suggested that the probability of adverse outcomes was lower with the pharmaco-invasive approach compared to facilitated PCI [293]. Routine early PCI after fibrinolysis significantly reduces reinfarction and the composite of death and reinfarction at 30 days, with benefits sustained at 12 months and no significant increase in major bleeding [294]. The greatest benefit is achieved when PCI is performed as soon as possible after fibrinolysis, without shifting to facilitated approaches [295]. Rescue PCI for failed fibrinolysis reduces reinfarction but does not impact mortality [296]. For people in hospitals without PCI capability, pathways should support early transfer for angiography when indicated [297]. Evidence supporting the recommendations
Practice points
- Perform regular ECG monitoring following fibrinolytic therapy according to local protocols, continuing until the person is pain-free and for at least 60–90 minutes post-fibrinolysis.
- Recognise failed reperfusion in people treated with fibrinolytic therapy if they exhibit any of the following:
- ongoing ischaemic chest pain
- ≤50% ST recovery on an ECG performed 60–90 minutes after fibrinolysis
- ongoing haemodynamic instability.
First Nations peoples
- Ensure that education about ongoing management is culturally appropriate and includes the importance of transferring to a PCI-capable centre when necessary.
- Provide cultural and language support before, during and after transfer, ensuring family and cultural considerations are addressed by appropriately trained staff.
People living in regional and remote areas
- Establish formal care pathways to facilitate timely and efficient transfer between non-PCI-capable centres and PCI-capable centres, often located in metropolitan areas. These pathways should address logistical challenges and ensure seamless continuity of care.
- Consider an additional half-dose of fibrinolytics with caution if fibrinolysis fails and timely transfer for rescue PCI is not feasible. However, this practice point is based on limited evidence from a single prospective trial with limited applicability to contemporary remote Australian settings [298].
Acute management of NSTEACS
Risk stratification for people with confirmed NSTEACS
Recommendations
Recommendations
Evidence supporting the recommendations
Evidence supporting the recommendations
Assessment of the short- and longer-term risk of death and recurrent ischaemic and bleeding events in people admitted with ACS can guide the need for, and timing of, invasive management. Risk assessment can also guide selection and duration of antithrombotic therapy. Clinical assessment and objective tools may both contribute to risk stratification in people with confirmed NSTEACS.
Clinical risk assessment
A subset of people present with factors that are associated with a high risk of short-term mortality, including haemodynamic instability/cardiogenic shock, life-threatening arrhythmias, mechanical complications of MI, acute heart failure clearly related to NSTEACS, and/or ongoing symptoms in the presence of high-risk ECG changes. These changes may include ST-segment depression >1 mm in more than six leads, STE in aVR and/or V1, Wellens criteria or recurrent intermittent STE (see Initial ECG assessment). An early invasive management strategy is recommended for these individuals.
In the absence of these very high-risk criteria, clinical risk assessment performs poorly compared with objective risk tools in determining prognosis.
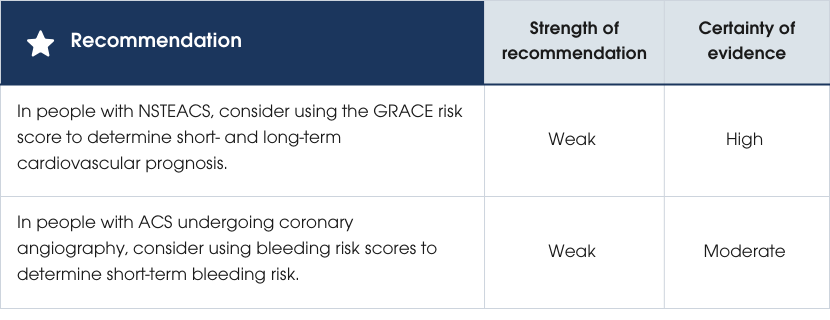
Objective risk prediction for ischaemic outcomes
The Global Registry of Acute Coronary Events (GRACE) risk score is a more accurate predictor of prognosis in NSTEACS compared to the thrombolysis in myocardial infarction (TIMI) risk score or subjective clinical assessment [21, 283, 299–306]. A stronger predictor of 30-day death or MI is baseline hs-cTn levels [307].Risk prediction for bleeding outcomes
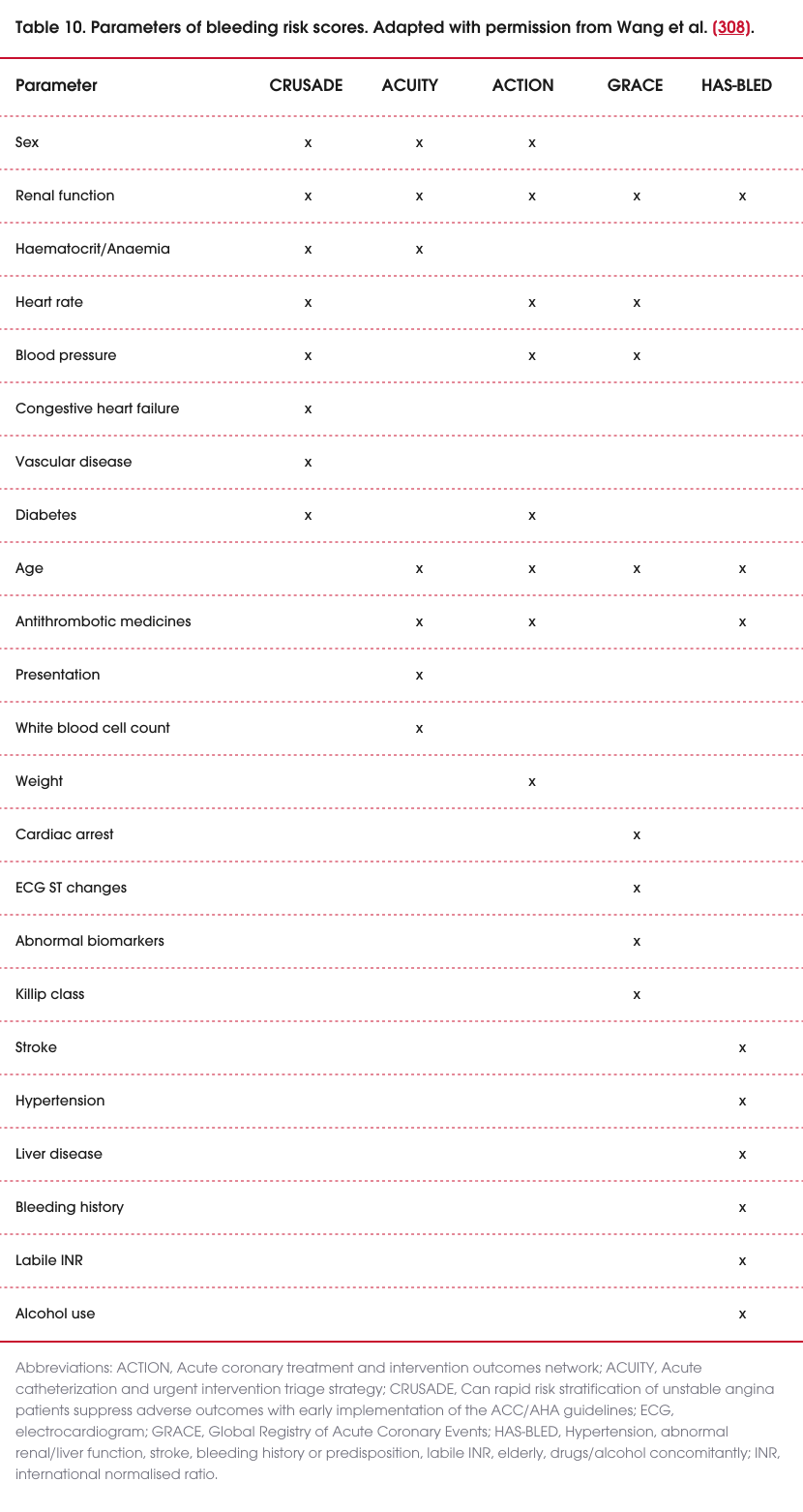
Major bleeding in hospital is associated with increased mortality, and a range of scores have been developed to predict this outcome among people presenting with ACS. The parameters of bleeding risk score is presented in Table 10. A comparison of scoring systems reported that acute coronary treatment and intervention outcomes network (ACTION) was the most accurate at predicting outcomes, followed by can rapid risk stratification of unstable angina patients suppress adverse outcomes with early implementation of the American College of Cardiology/American Heart Association guidelines (CRUSADE) and acute catheterization and urgent intervention triage strategy (ACUITY) [308]. The Academic Research Consortium high bleeding risk (ARC-HBR) score is an alternative pragmatic approach recommended by European guidelines [309].These scores were developed in populations with a high prevalence of coronary angiography and DAPT use. While they may be considered when choosing procedural and antiplatelet strategies, their impact on outcomes has not been established.
Practice points
- Recognise that GRACE risk scores were developed before the introduction of hs-cTn assays. However, most people identified as high risk by the GRACE score are similarly identified using hs-cTn testing alone.
- Prioritise bleeding risk over ischaemic risk when making decisions about the duration of DAPT in people at risk of both bleeding and ischaemic events, as suggested by observational data. Refer to sections Antiplatelet therapy in the acute phase and Anticoagulant therapy in the acute phase [310]
Women
- Use the GRACE 3.0 score for risk assessment in women with NSTEACS, as it provides a more accurate estimation of mortality risk than the GRACE 2.0 (which underestimates mortality) and helps address sex inequalities in risk stratification [311].
Older adults
- Consider that the GRACE risk score heavily weights age and does not account for characteristics common in older adults, such as frailty, multimorbidity, polypharmacy and cognitive dysfunction, which can contribute to higher risk scores [312].
- Assess frailty in older adults, as it is independently associated with adverse outcomes and increased bleeding risk [313]. Use validated frailty assessment tools to guide management decisions [314–317].
- Consider a conservative management approach in older adults, even if they are deemed high risk for ischaemic events based on objective scoring, particularly when frailty and bleeding risk are significant concerns.
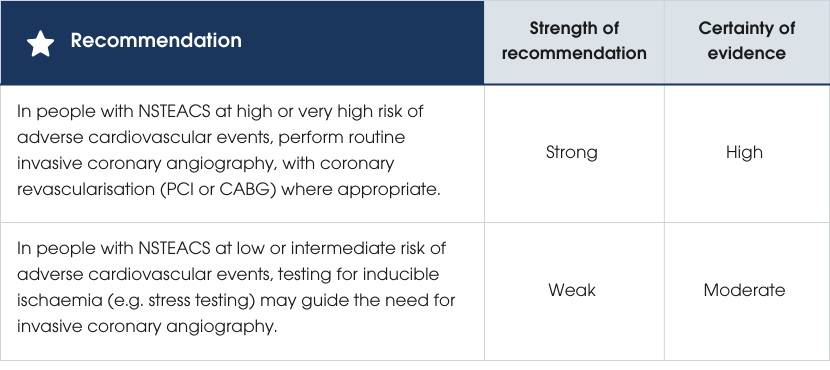
Routine versus selective invasive management for NSTEACS
Recommendations
Recommendations
Evidence supporting the recommendations
Evidence supporting the recommendations
Criteria used to identify people with confirmed NSTEACS at high and very high risk of adverse cardiovascular events or death:
| Risk category | Criteria |
|---|---|
| High risk |
|
| Very high risk |
|
Practice points
- Consider the goals of therapy, individual preferences and the impact of major comorbidities when deciding on the appropriateness of a routine invasive approach for people with NSTEACS.
- Use anatomical imaging with CTCA instead of functional testing to exclude or define CAD in people with NSTEACS who:
- are not at high or very high risk of adverse cardiovascular events
- do not have known CAD
- present with an unclear NSTEACS diagnosis, as detailed in Section Further diagnostic testing for people with suspected ACS [328, 329] .
This approach can enhance diagnostic clarity and tailor management strategies effectively.
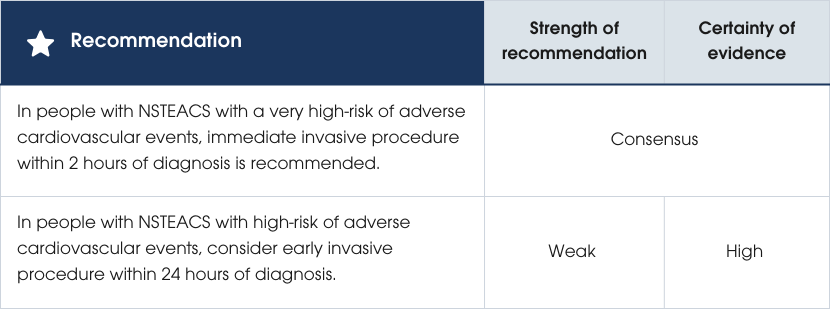
Timing of invasive management for NSTEACS
Recommendations
Recommendations
Evidence supporting the recommendations
Studies on the timing of invasive coronary angiography in NSTEACS, comparing early intervention (e.g. within 24 hours) with delayed intervention (e.g. 2–3 days), found no overall benefit in mortality, MI or stroke when applied to all participants without considering individual risk. Risk stratification should guide timing decisions NSTEACS [330–332] . Evidence supporting the recommendations
For unstable or very high-risk individuals, immediate angiography (within two hours) is recommended based on poor outcomes without intervention, although this is supported by expert opinion rather than robust evidence (Figure 13).
In people at high risk (e.g. GRACE score >140), early intervention reduced death, MI and stroke at 6 months compared to delayed strategies (14% vs 21%), without increasing major bleeding. Mortality benefits were also observed in those with elevated biomarkers, diabetes, GRACE score >140, or aged ≥75 years, but evidence for specific risk-treatment interactions is limited. Data using hs-cTn-based GRACE scores remain unavailable [330, 333] .
Figure 13 Timing of invasive management for NSTEACS. Abbreviations: ECG, electrocardiogram; GRACE, Global Registry of Acute Coronary Events; hs-cTn, high-sensitivity cardiac troponin; MI, myocardial infarction; NSTEACS, non-ST-segment elevation acute coronary syndromes.
Evidence supporting the recommendations
Studies consistently show that radial access reduces mortality (1.6% vs 2.1%) and major bleeding (1.5% vs 2.7%) compared to femoral access in people with NSTEACS [271–275]. A radial-first approach is recommended unless there is a lack of operator experience or there are contraindications. Intravascular imaging (IVI)-guided PCI, using optical coherence tomography or intravascular ultrasound (IVUS), reduced target lesion failure by lowering the risks of cardiac death, target vessel MI and target lesion revascularisation, in addition to reducing all MI and all-cause death compared to angiography-guided PCI. Outcomes were similar for optical coherence tomography- and IVUS-guided procedures [334]. The benefit of IVI-guided PCI was of similar or greater magnitude in people with ACS, particularly for complex lesions and higher-risk individuals (e.g. bifurcations, calcifications, long lesions or diabetes) [335]. However, recommendations for IVI-guided PCI should be tailored and not applied universally to all PCI procedures. Evidence supporting the recommendations
Practice points
Women
- Use a radial-first approach where possible for coronary interventions in both women and men, as it is associated with reduced complications and improved outcomes.
- Consider a routine invasive approach for women with NSTEACS, as it has demonstrated benefits. Do not overlook the disparity in care, as observational data show women are less likely than men to receive an invasive strategy or radial access [322, 336].
Older adults
- Consider an invasive strategy over an initial conservative approach in older adults with NSTEACS if they do not have frailty, multimorbidity or cognitive dysfunction based on objective assessment. The evidence to consider is:
- Of five trials on invasive management in older adults with NSTEACS (mostly ≥75 years), four found no benefit in primary endpoints, but one showed reduced MI and urgent repeat revascularisation with a routine invasive strategy [337–342].
- For older individuals with MI and multivessel disease, physiology-guided complete revascularisation showed benefits, although frailty data were not provided [343, 344].
- Meta-analyses suggest that an invasive strategy likely reduces MI and recurrent revascularisation compared to conservative management, with observational studies suggesting a survival benefit and randomised trials showing a trend towards improved survival. However, this must be balanced against an increased risk of bleeding [344–346].
- A small trial of frail individuals aged over 70 years (mean age 86) with NSTEACS found no benefit from an initial invasive approach [347].
Individualise treatment decisions for older adults, balancing the potential for improved outcomes with the risks of complications, especially bleeding.
First Nations peoples
- Ensure information about transfers or invasive management is provided with the assistance of First Nations health practitioners or Aboriginal liaison officers.
- Communicate in the person’s preferred language when required to enhance understanding and informed decision-making.
- Recognise that First Nations peoples from regional areas are less likely to receive angiography compared to non-Indigenous counterparts [348].
- Be vigilant of barriers to equitable care, including:
- inadequate cultural competency among healthcare providers
- perceptions about medicine compliance
- delayed transfers to PCI-capable hospitals
- insufficient family and community engagement by clinicians [52].
Evidence supporting the recommendations
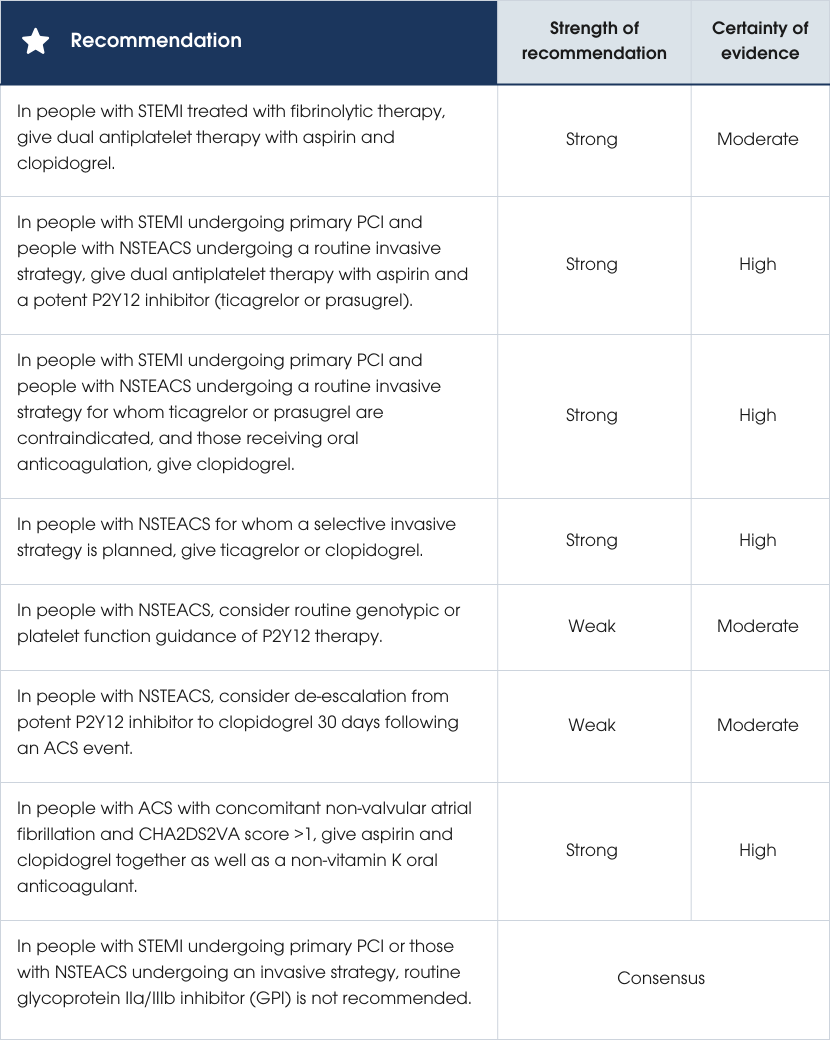
People with NSTEACS can defer P2Y12 inhibitor loading until after coronary angiography, provided that angiography is performed within recommended timelines (Section Further diagnostic testing for people with suspected ACS) [357, 358]. In STEMI undergoing primary PCI, pretreatment with a P2Y12 inhibitor may be considered if the working diagnosis is certain, but if pretreatment is not given, all people should receive a P2Y12 inhibitor loading dose at the time of PCI (see Supplementary Material B3) [359]. Genetic or platelet function guidance to tailor P2Y12 therapy has not consistently demonstrated net clinical benefit, but ongoing studies may clarify its role [360–364].
Evidence supports de-escalation from potent P2Y12 inhibitors to clopidogrel one month post-ACS to reduce bleeding risk, without clear evidence of increased ischaemic events [365–368]. For people requiring concomitant OAC, particularly with non-valvular atrial fibrillation, initial short-term triple therapy (aspirin, clopidogrel and an OAC) followed by dual therapy (OAC plus clopidogrel) effectively reduces bleeding risk [369–371]. The recommended discontinuation intervals prior to non-emergency cardiac surgery for ACS are five days for clopidogrel, three days for ticagrelor and seven days for prasugrel [372].
Finally, routine IV GPI inhibitor use is not recommended in primary PCI or in routine invasive strategies for NSTEACS, although bailout use may be considered in select high-thrombus-burden circumstances [373–379]. Further evidence to support the recommendations is provided in the Comprehensive Guideline, and further details on switching strategies, loading protocols and timing of administration are provided in the and the Supplementary Material B3.
Practice points
- Aspirin sensitivity: In the event of aspirin sensitivity, risk assessment and consideration of desensitisation should be made using a standardised protocol to achieve adequate antithrombotic therapy [380].
- Selection of platelet P2Y12 inhibitor therapy: Prasugrel has Therapeutic Goods Administration (TGA) approval but is not currently available in Australia. Exercise care regarding timing and dosing of P2Y12 inhibitors when switching between these agents to ensure their effectiveness is maintained and the bleeding risk minimised. For guidance on switching strategies, (see Supplementary Material B3).
- Timing of platelet P2Y12 inhibitor administration in STEMI: Deferring the administration of the P2Y12 inhibitor until after the coronary anatomy is known is reasonable when the diagnosis of STEMI is uncertain or if there is a clinical suspicion of need for urgent cardiothoracic surgery (e.g. left main ischaemia pattern on ECG).
- Timing of platelet P2Y12 inhibitor initiation in NSTEACS: Decisions regarding timing of initiation of P2Y12 inhibitor in relation to invasive angiography may be institution-dependent and need to be clearly defined and communicated effectively between emergency and inpatient services.
- Combining P2Y12 inhibition with anticoagulation: In people with ACS with an indication for vitamin K antagonist (e.g. mechanical heart valve), use aspirin with clopidogrel rather than ticagrelor or prasugrel to reduce the risk of bleeding. Target international normalised ratios (INRs) should be at the lower therapeutic range (e.g. 2.5–3 for mechanical mitral valves).
- IV GPI administration: Bailout GPI may be considered in people at high ischaemic risk such as high thrombus burden, no-flow or slow-flow.
- Discontinuing platelet P2Y12 inhibitor prior to CABG: In people with NSTEACS with planned CABG, do not administer P2Y12 inhibitor within three days of surgery for ticagrelor, five days for clopidogrel or seven days for prasugrel.
- Selection of GPI therapy: Tirofiban is the only GPI marketed in Australia, while eptifibatide and abciximab can be obtained through the TGA’s Special Access Scheme.
- Discontinuing IV GPI in thrombocytopaenia: Glycoprotein IIb/IIIa inhibition is not recommended in people with thrombocytopaenia (platelet count <150,000/mL) and should be suspended immediately if platelet count falls below this level or drops by 50% or more from baseline.
- Discontinuing IV GPI prior to CABG: In people undergoing CABG, discontinuation of short-acting GPI (eptifibatide and tirofiban) for four hours and abciximab for 12 hours before surgery is recommended to reduce the risk of bleeding and transfusion [381–383].
Evidence supporting the recommendations
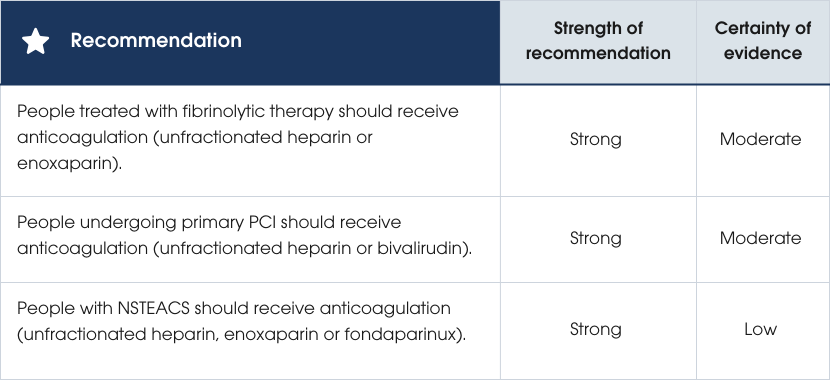
Anticoagulation is recommended for people with ACS, whether managed with fibrinolytic therapy, primary PCI or a NSTEACS strategy. In fibrinolysis, the GUSTO trial showed the lowest mortality among people with STEMI who received tPA and IV heparin [384]. ASSENT-3 demonstrated fewer ischaemic events with tenecteplase plus enoxaparin (30 mg IV bolus followed by 1 mg/kg subcutaneously twice daily) compared with IV heparin [385]. For primary PCI, early studies found bivalirudin had similar efficacy but lower bleeding risk than unfractionated heparin (UFH) when GPI inhibitors were used routinely [280, 386, 387]. More recently, the BRIGHT-4 trial (93% radial access; bailout, not routine, GPI) reported that bivalirudin significantly reduced mortality and major bleeding compared with UFH (0.7 units/kg) [388]. Bivalirudin can therefore be considered instead of UFH in people undergoing primary PCI for STEMI, factoring in differences in cost and experience with administration. Bivalirudin should be used instead of UFH in people with heparin-induced thrombocytopaenia. In NSTEACS, Early trials showed that UFH reduces MACE without increasing bleeding [389-391], forming the basis for anticoagulation in higher-risk ACS. Although low molecular weight heparin (LMWH) on background therapy with aspirin can also reduce MACE [392], a larger trial showed there was no difference in the ischaemic endpoint of death and MI but significantly increased bleeding (commonly related to femoral access) with LMWH in settings of peoples on DAPT, early angiography and frequent GPI use [393]. Contemporary meta-analyses suggest no ischaemic advantage of bivalirudin over UFH, especially with radial access [394]. Finally, fondaparinux halves major bleeding compared with LMWH in people on DAPT without compromising efficacy (there were high rates of angiography in these trials) [392, 395]. Evidence supporting the recommendations
Practice points
Anticoagulant treatment with fibrinolytic therapy
- Omit IV bolus of enoxaparin in people >75 years receiving fibrinolysis and enoxaparin.
- Enoxaparin is recommended over UFH unless there is severe kidney impairment (eGFR <30 ml/min) [396, 397] .
Anticoagulant therapy with primary PCI
- In people requiring PCI with a history of heparin-induced thrombocytopaenia, consider bivalirudin as an alternative to UFH. Outcomes with bivalirudin are optimised when followed by a high dose post-PCI infusion (1.75 mg/kg/hr) for 2–4 hours [398].
Anticoagulant therapy in NSTEACS
- In people treated with fondaparinux undergoing coronary angiography and/or PCI, standard dose UFH is recommended at the time of the procedure to reduce the risk of guiding-catheter thrombosis [399, 400].
- In people receiving LMWH in whom femoral access for coronary angiography is planned, it is common practice to omit the morning dose of enoxaparin to minimise access-related bleeding complications [401].
- Parenteral anticoagulants can be ceased following PCI. In people who do not undergo PCI, UFH may be ceased at 48 hours and fondaparinux or LMWH at six days following presentation in the absence of other indications [393, 399].
Anticoagulant use in people already receiving warfarin or direct oral anticoagulants
In people with continued indications for oral anticoagulants (atrial fibrillation and CHA2DS2VA score >1, mechanical heart valves or recurrent venous thromboembolism), do not cease this treatment.
In people with NSTEACS undergoing invasive management, wherever possible a brief washout period from the effects of oral anticoagulants (OACs) is desirable. This is to reduce the risk of potential bleeding complications among those who may require femoral access or resulting from additional anticoagulation during the procedure. The suggest washout period is 24 hours for people on direct oral anticoagulants (DOACs) with normal renal function and 48 hours for those with impaired renal function. For people on warfarin, an INR of <2.0 is recommended when using the radial approach and <1.5 when using the femoral approach.
- There are no randomised studies evaluating strategies for early anticoagulation in people with ACS who are already taking warfarin or DOACs. Guidance for these people is derived from expert opinion [402].
Acute management of ACS with cardiac arrest and/or cardiogenic shock
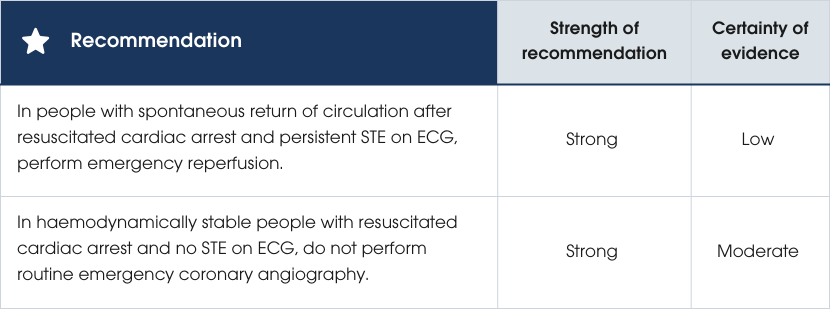
ACS with cardiac arrest
Recommendations
Evidence supporting the recommendations
Practice points
- In people with STEMI and resuscitated cardiac arrest, primary PCI is the preferred reperfusion strategy. Fibrinolysis may be considered if primary PCI is unavailable. However, evidence is lacking with potential for harm in cardiac arrest that is refractory, prolonged and/or traumatic [256, 264, 408].
- In people with STEMI and resuscitated cardiac arrest, the decision for primary PCI should factor in treatment futility. For instance, advanced age, presence of severe metabolic acidosis and/or no return of spontaneous circulation for an extended period are associated with a low likelihood of meaningful long-term survival [409].
Evidence supporting the recommendations
Treatment of coronary microvascular disease in ACS with cardiogenic shock
In NSTEACS with coronary microvascular disease (MVD) and cardiogenic shock, culprit-lesion-only PCI reduced the composite of death or renal replacement therapy compared to multivessel PCI, driven by lower mortality [410]. For STEMI with cardiogenic shock, non-culprit lesion PCI during the initial procedure increased death and renal failure risk. Therefore, in the presence of cardiogenic shock, PCI of non-IRAs should not be performed at the time of the index procedure; staged PCI is recommended for complete revascularisation [410–412].Haemodynamic support devices in MI and cardiogenic shock
Routine intra-aortic balloon pump use in MI with cardiogenic shock increases bleeding without survival benefit [413–415]. Early venoarterial extracorporeal membrane oxygenation (VA-ECMO) showed no mortality benefit but increased major bleeding and peripheral vascular complications [416, 417]. Percutaneous left ventricular assist devices reduced mortality but increased bleeding, vascular complications and haemodynamic shock in people with severe left ventricular impairment [418].Practice points
- Consider intra-aortic balloon pump in select cases – for example, where there are mechanical complications (ventricular septal rupture, mitral regurgitation or free ventricular wall rupture) – and/or as bridging to heart transplant or left ventricular assist devices.
- Consider mechanical support including VA-ECMO on a case-by-case basis as rescue or bridging therapy. Such support can also be considered for treatment of intractable ventricular tachyarrhythmias, in consultation with a multidisciplinary team.
- Consider left ventricular assist devices in people with STEMI and cardiogenic shock on a case-by-case basis, given the selected population enrolled and the complication rate in the DanGer Shock trial.
- In people with ACOMI and cardiogenic shock, where PCI is unavailable, consider fibrinolysis with a plan for subsequent angiography (see recommendations in Section Ongoing management of fibrinolytic-treated people) [419].
Evidence supporting the recommendations
Evidence supporting the recommendations
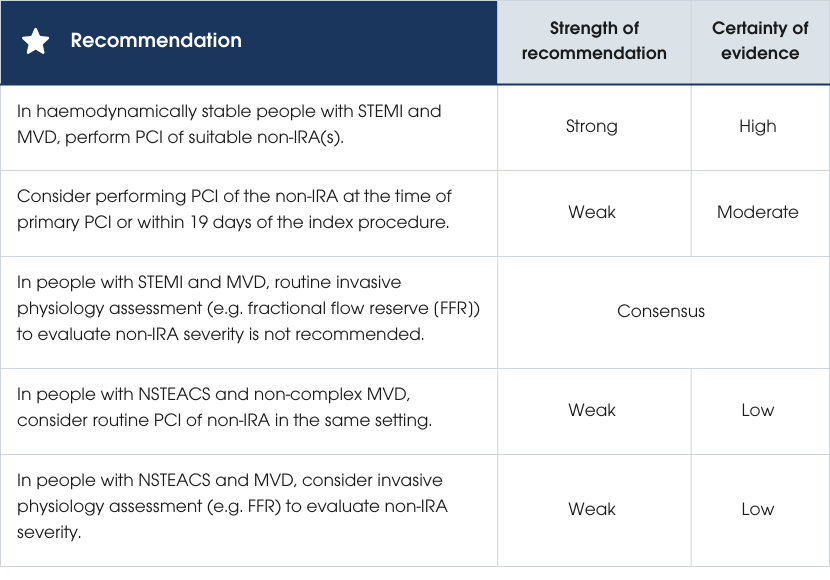
Treatment of MVD in STEMI
Complete revascularisation in STEMI, MVD and without cardiogenic shock reduces cardiac death, MI and repeat revascularisation compared to IRA-only PCI. Immediate revascularisation at the index procedure is superior to outpatient-staged PCI but its advantage over inpatient-staged PCI remains unclear. CABG may be preferred for complex MVD cases (Section Coronary artery bypass graft surgery in ACS) [289, 420–433].Treatment of MVD in NSTEACS
No trials specifically compare complete versus IRA-only PCI in NSTEACS. A meta-analysis of observational studies suggests higher short-term risk but improved long-term outcomes with complete revascularisation [434].Invasive physiology to evaluate the non-IRA in STEMI or NSTEACS and MVD
In STEMI with MVD, angiography-guided PCI is effective and may outperform physiology-guided approaches for non-IRA lesions [343, 435, 436]. In NSTEACS, physiology-guided PCI may reduce unnecessary revascularisation but outcomes are inconsistent [437, 438]. Among older adults, physiology-guided PCI improves outcomes with no difference in safety outcomes [439].For guidance on management of MVD in people with ACS, refer to Figure 14.

Figure 14 Management of multivessel disease in people with ACS. Abbreviations: ACS, acute coronary syndromes; FFR, fractional flow reserve; IRA, infarct-related artery; NSTEACS, non-ST-segment elevation acute coronary syndromes; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction.
Practice points
Treatment of non-IRAs
- In people with STEMI and MVD, with unknown renal function, inpatient PCI as a staged rather than immediate procedure may be preferable if complex MVD is present or operator fatigue precludes same setting multivessel PCI.
- In people with NSTEMI and MVD, timing for complete revascularisation should consider factors such as the presence of cardiogenic shock, lesion complexity and risk of contrast nephropathy.
- While a benefit of FFR-guided over angiography-guided complete revascularisation has not been conclusively shown, it is reasonable to use FFR in intermediate (50–69%) non-infarct-related stenoses [435].
- In people with ACS and complex MVD, a multidisciplinary heart team approach to the revascularisation strategy is recommended. Management of people with ACS and complex MVD should be guided by multidisciplinary heart team discussions incorporating person-based (e.g. age, frailty, infarct size, personal preference) and lesion-based (e.g. location, severity and complexity) factors.
Evidence supporting the recommendations
Perioperative mortality after mechanical complications of STEMI remains high [440] . Few percutaneous or medical treatments are available, and urgent surgery is often the best option. A haemodynamically unstable person may require interim mechanical circulatory support. Performing CABG at the time of surgery for a mechanical complication of STEMI is based on small retrospective series with no randomised trial data [441]. Evidence supporting the recommendations
Practice points
- In people with STEMI where PCI cannot be performed, consider emergency CABG if there is ongoing ischaemia and a large area of jeopardised myocardium.
- Overall, 4–10% of people with NSTEACS will require CABG [442]. When deciding between PCI and CABG, consider comorbidities, fitness for major surgery and coronary anatomy. A multidisciplinary team should be involved in decision-making.
- In people with ACS and MVD where CABG has been chosen as the complete revascularisation strategy, performing CABG at day 1 to day 7 (compared to day 0 or >7 days) after diagnosis has lowest risk of mortality [443].
- In people with ongoing ischaemia or haemodynamic instability with an indication for CABG, do not delay urgent surgery due to antiplatelet exposure.
Evidence supporting the recommendations
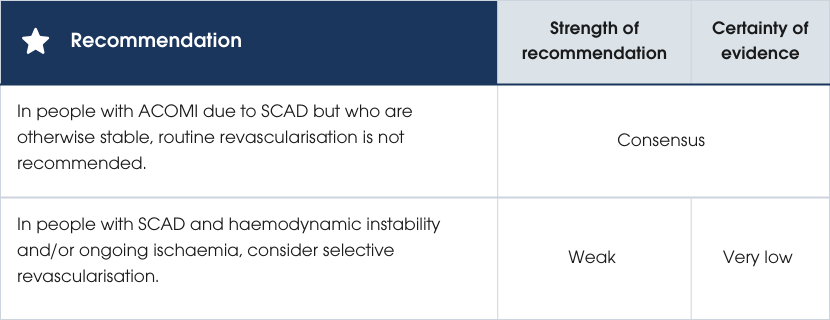
As there are no randomised controlled trials (RCTs) to guide therapy, recommendations in SCAD are based on observational studies or expert opinion [444]. Intervention is challenging, and routine revascularisation is not recommended as it has been associated with several complications. These include iatrogenic dissection, wiring of the false lumen, propagation of the intramural haematoma, acute vessel closure and stent or graft failure [445–447]. However, in a subgroup of people with SCAD who have significant ongoing ischaemia and haemodynamic compromise, urgent revascularisation with PCI or CABG may be required [175, 448–450]. Evidence supporting the recommendations
Myocardial infarction with non-obstructive coronary arteries
In people with myocardial infarction with non-obstructive coronary arteries (MINOCA), it is important to exclude alternative diagnoses [451]. Consider cardiac MRI in all people with MINOCA where the underlying cause is not obvious. Once the underlying cause has been established, manage people with MINOCA according to relevant disease-specific guidelines [2]. In all people with evidence of coronary atherosclerotic disease and/or risk factors, consider initiating secondary prevention measures (even if the underlying cause of MINOCA cannot be determined) [452].MI due to oxygen supply/demand mismatch without acute coronary occlusion
No trials have examined the benefits of a routine invasive strategy in people with MI due to oxygen supply/demand mismatch without acute coronary occlusion [453]. Whether competing risks from non-cardiac conditions obscure the benefits of invasive management – and at what level of competing risk this occurs – remains uncertain. All available evidence demonstrates that people with MI due to oxygen supply/demand mismatch without acute coronary occlusion experience higher all-cause mortality than people with MI with acute coronary occlusion. This is, in part, related to associated non-coronary competing risks [453].In the absence of any trial evidence, angiography with a view to revascularisation may be considered if there is ongoing ischaemia or haemodynamic compromise despite adequate treatment of the underlying acute stressors that provoked the MI due to oxygen supply/demand mismatch without acute coronary occlusion (Section Administration of fibrinolytic therapy and Table 9).
Echocardiography
Left ventricular (LV) dysfunction is an important determinant of prognosis following ACS, and its detection should guide further evidence-based therapies [454]. Echocardiography to evaluate regional and global LV function, and to identify other cardiac pathology, should be performed during hospitalisation. If echocardiography is not possible, consider other aspects suggestive of LV dysfunction, including clinical signs/symptoms, and ECG, chest X-ray and biomarker features [454].Duration of cardiac monitoring
Cardiac monitoring plays a pivotal role as an adjunct therapy in the management of ACS. Continuous cardiac monitoring has become a firmly embedded standard of practice despite the absence of evidence from RCTs [455]. Clinical assessment for the risk of life-threatening arrhythmias should be individualised based on known associated risk factors: arrhythmias, ongoing symptoms, reduced LV function (LV ejection fraction [LVEF] <40%), failed coronary reperfusion, haemodynamic instability and complications of PCI (side branch occlusion, unsealed dissection, embolisation).Practice points
- In people with ACS, initiate cardiac monitoring immediately, with ST-segment ischaemia monitoring where available. Continue uninterrupted for a minimum of 24 hours.
- People with ACS post-PCI should be monitored, with ST-segment ischaemia monitoring where available, continuously and uninterrupted for 24 hours.
- Re-evaluate the need for continuous ECG monitoring every 24 hours.
- Educate staff regarding proper skin preparation, assessment of skin turgor and ECG electrode replacement every 24 hours, as this reduces inappropriate alarms [456, 457].
- Further guidance regarding cardiac monitoring can be found on the Agency for Clinical Innovation website (aci.health.nsw.gov.au/cardiac) [455].
1. Assessment and diagnosis
3. Recovery and secondary prevention