Australian clinical guideline for diagnosing and managing acute coronary syndromes 2025

Assessment of people with suspected ACS
Initial ECG assessment
Biomarkers
Risk assessment and clinical decision pathways for suspected ACS
Initial therapeutic management
Further diagnostic testing for people with suspected ACS
Role of rapid access chest pain clinics
Re-presentation with symptoms
Primary care and regional and remote presentations
Discharge planning and advice
 1. Assessment and diagnosis
1. Assessment and diagnosis
Assessment of people with suspected ACS
Acute chest pain is a relatively common emergency department (ED) presentation, yet only a minority of people will be diagnosed with ACS [22, 23]. Among those presenting with acute chest pain to the ED in whom ACS is suspected, <5% will have STEMI, 5–10% NSTEMI, 5–10% UA, 15–20% other cardiac conditions, and 50–60% non-cardiac conditions [23–26]. Most people will therefore require further follow-up to assess and diagnose their condition [27].Assessment for ACS includes:
- history and physical examination
- ECG
- troponin testing.
These are required to diagnose as well as inform risk assessment and help guide the location and timing of further investigations, management and follow-up. Rapid identification and diagnosis of ACS is crucial as treatments are often time sensitive and earlier intervention improves outcomes.
Assessing a person’s relative risk for ACS is the key initial goal, rather than achieving a conclusive diagnosis of ACS, which may not always be possible at the time (Section Risk assessment and clinical decision pathways for suspected ACS).
Assessment in suspected ACS should:
- identify people with acute coronary occlusion myocardial infarction (ACOMI) (STEMI and STEMI equivalents)
- identify people with NSTEMI
- identify people with UA at high risk for 30-day MACE
- identify people with underlying CAD in whom ACS is not confirmed.
Assessment for people with suspected ACS within an ED setting is described below. Specific guidance for people presenting in regional/remote and primary care settings is given in Section Primary care and regional and remote presentations.
Initial assessment summary
In people presenting with symptoms suggestive of ACS, the following steps are recommended (see Practice points for setting considerations):
- Vital signs including blood pressure, heart rate, respiratory rate and peripheral oxygen saturation should be recorded.
- An ECG should be reviewed by a clinician experienced in ECG interpretation to examine for evidence of ACOMI within 10 minutes of presentation (Section Initial ECG assessment).
- If a diagnosis of ACS is considered likely, further investigations, including troponin testing, should be performed (Section Biomarkers and Section Risk assessment and clinical decision pathways for suspected ACS).
- Promptly identify people with ACOMI suitable for urgent reperfusion.
- People without symptoms of ACOMI but who have ECG evidence of cardiac ischaemia, and/or people who are otherwise stratified as high risk of index MI or 30-day MACE (Section Clinical decision pathways) should have continuous cardiac monitoring.
- In people who are symptomatic and/or haemodynamically compromised, ECGs should be performed at (a minimum of) 15-minute intervals until the symptoms have resolved.
- Additional ECGs should be performed if the person’s symptoms reoccur, there are changes in character or a change in their clinical condition.
- If there is no evidence of ACOMI on ECG, a targeted history and physical examination should be performed and differential diagnoses considered, particularly time-critical emergencies, including ruptured aortic aneurysm, pulmonary embolism or pneumothorax (Table 2).
- A chest X-ray may be useful in identifying some other causes, including pneumonia or pneumothorax, and to assess for cardiac size or evidence of cardiac failure. Acquiring a chest X-ray should not delay urgent reperfusion. This guideline does not apply to these other time-critical emergencies [28, 29].
- If the ECG is normal, and symptoms are clearly attributable to a non-cardiac cause, this guideline no longer applies. Clear communication is essential. Explaining that they do not have ACS may reduce a person’s and their carers’ anxiety (Section Discharge planning and advice).
| Cardiac: ACS | AMI, unstable angina |
| Cardiac: Other | Stable angina, myopericarditis, tachyarrhythmia, hypertensive emergencies, severe aortic stenosis, Takotsubo cardiomyopathy, cardiac trauma |
| Pulmonary | Pulmonary embolism, pneumothorax (including tension), infection (pneumonia, bronchitis), pleuritis |
| Vascular | Aortic dissection, expanding aortic aneurysm, sickle cell crisis |
| Gastrointestinal | Oesophagitis, reflux, spasm, rupture, peptic ulcer disease, pancreatitis, cholecystitis and biliary disease |
| Other | Musculoskeletal disease (including costochondritis, trauma), anxiety disorder, infectious disease (including herpes zoster) |
Abbreviations: ACS, acute coronary syndromes; AMI, acute myocardial infarction.
The following sections describe the subsequent processes of assessment and diagnosis to be followed if ACS is suspected.
History of the presenting complaint
After assessing vital signs and ECG, obtain a focused medical history, including symptoms consistent with MI, onset and timing, associated symptoms and ACS risk factors. Use translator services and culturally competent healthcare workers as needed to address language, cultural or hearing barriers [30] (Practice points: First Nations peoples).Chest pain, angina equivalents and associated symptoms
Chest pain is the most common symptom of ACS, yet it is not always present. Chest pain due to myocardial ischaemia is often described as substernal discomfort or pressure, which may radiate to the neck, arms or jaw. The pain is often exacerbated by exertion and relieved following 15–20 minutes of rest [31]. Chest pain due to MI or UA generally occurs at rest. People may also refer to a discomfort, pressure or heaviness and deny pain. In this guideline, discomfort, pressure and heaviness are included under the umbrella term of chest pain.Descriptions of myocardial ischaemic pain vary according to sex, ethnicity and culture. The description of the pain may help in determining whether the person’s presentation is consistent with myocardial ischaemia (Figure 3).
A response or lack of response to treatment (such as glyceryl trinitrate, standard analgesia or antiacids) should not be used as a diagnostic criterion for ACS (Section Initial therapeutic management) [32].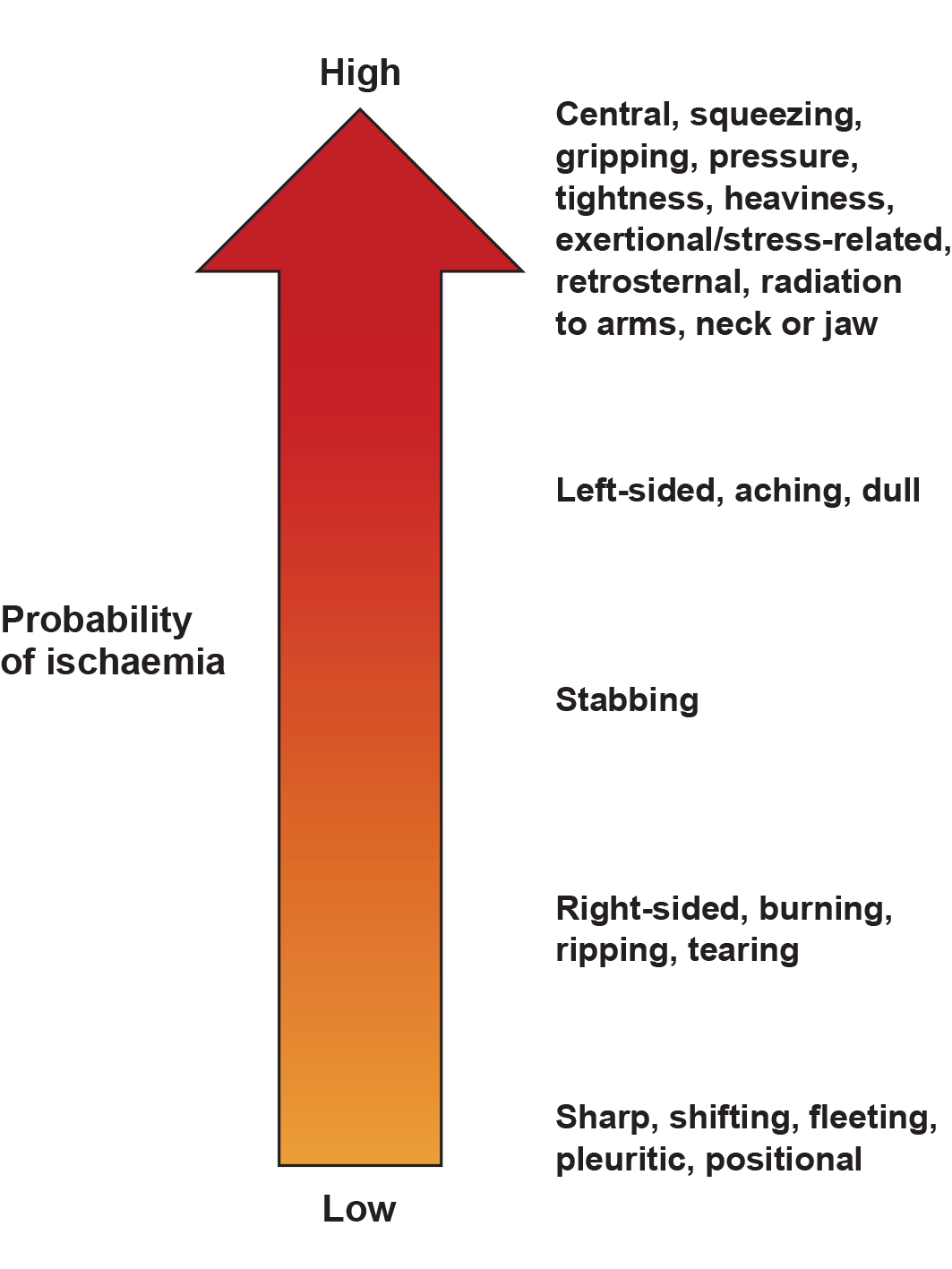
Figure 3 Probability of cardiac ischaemia based on commonly used descriptors of chest pain.
Shortness of breath, fatigue, nausea, diaphoresis or vomiting are relatively common associative symptoms of ACS. Women are more likely than men to report these symptoms (Practice points: Women). Some people, particularly older adults and people with diabetes, may not describe any chest pain or discomfort and report only associated symptoms (sometimes referred to as angina or chest pain equivalents).
The terms typical and atypical have previously been used to describe cardiac ischaemic symptoms. However, given their wide variation, cardiac, possible cardiac or non-cardiac symptoms are now recommended terms (Practice points: Women and Older adults) [27].Factors associated with MI types
The risk factors for the different types of AMI are listed in Table 3. An absence of risk factors for CAD does not exclude ACS, which may present as either MI with acute coronary occlusion or MI due to oxygen supply/demand mismatch.
Factors associated with atherosclerosis[33, 34]:
|
Factors associated with SCAD[35]:
|
Factors associated with coronary embolism[36]:
|
Factors associated with coronary vasospasm[37-39]:
|
Factors associated with coronary microvascular dysfunction[40]:
|
Factors associated with oxygen supply/demand imbalance (+/- atherosclerosis)[1]:
|
Note: Takotsubo cardiomyopathy is not classified as MI and is not discussed in this guideline. Abbreviations: AIDS, acquired immunodeficiency syndrome; ASCVD, atherosclerotic cardiovascular disease; eGFR, estimated glomerular filtration rate; HIV, human immunodeficiency virus; MI, myocardial infarction; SCAD, spontaneous coronary artery dissection.
Practice points
Women
- Recognise chest pain as the most common symptom in women with ACS, occurring at similar rates to men. Be aware that associated symptoms in women may include jaw, neck, shoulder or back pain, fatigue, nausea, vomiting, dizziness, indigestion and shortness of breath [41–43].
- Remain vigilant to the risk of misdiagnosis in women presenting with ACS [44, 45]. Women are more likely than men to be misdiagnosed with non-cardiac pain and are more likely to experience delays in receiving life-saving procedures in hospital, and healthcare professionals should consider potential sex bias when interpreting symptoms [14, 46].
- Increase awareness of sex differences in ACS symptoms and presentation patterns. Clinician education and recognition of these differences may improve diagnosis and management of ACS in women.
- Consider SCAD as a potential cause of ACS, particularly in young to middle-aged women. Refer to (Section Treatment for SCAD) for management recommendations [33, 35].
Older adults
- Recognise that older age (>75 years) is an independent risk factor for ACS and other conditions with similar presentations. Comorbidities are also more prevalent in this age group, which may complicate diagnosis and management [47].
- Be aware that chest pain may not be the primary symptom of AMI in older adults. In this population, including those with STEMI and STEMI equivalents, angina equivalents (such as shortness of breath, fatigue, or dizziness) being commonly observed [48–51].
First Nations peoples
- Recognise that First Nations peoples with ACS are typically younger, have higher rates of cardiac risk factors, experience lower intervention rates and face poorer outcomes compared to non-Indigenous Australians. These disparities highlight the importance of tailored interventions and equitable care [17, 52, 53].
- Provide access to First Nations health practitioners, liaison officers and culturally appropriate interpreter services within hospitals. This facilitates accurate history-taking and improves the quality of care [17, 52].
- Incorporate culture-specific attitudes and values into health promotion tools and offer culturally appropriate pastoral care. These steps can help bridge cultural gaps and enhance engagement with First Nations peoples [17, 52].
- Prioritise education on cultural awareness, competency and safety for healthcare professionals. Such education has been shown to reduce unconscious bias and improve health outcomes for First Nations peoples [17, 52].
Initial ECG assessment
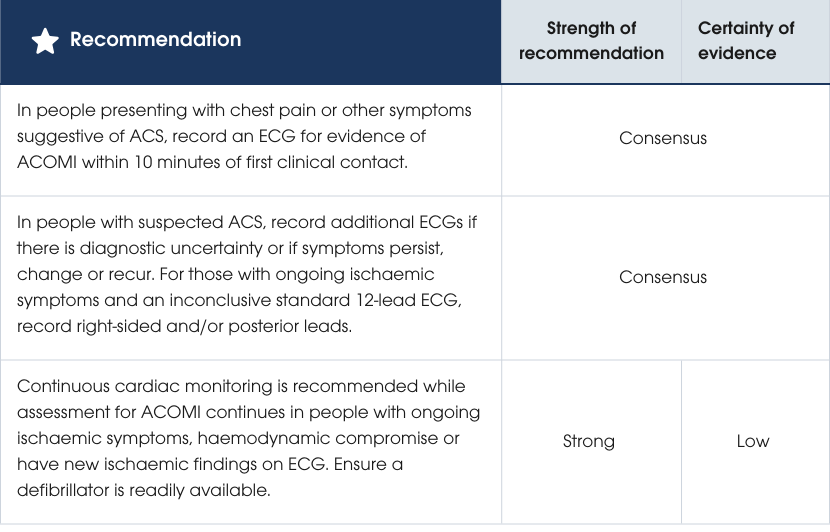
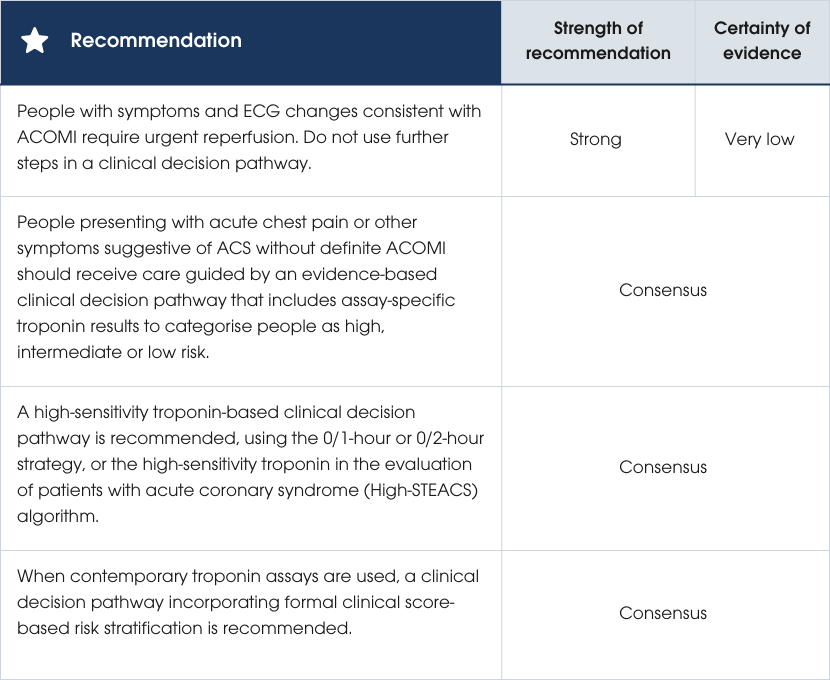
Recommendations
Recommendations
Evidence supporting the recommendations
The priority in screening for ACS is to identify ACOMI early to expedite reperfusion and improve outcomes. Urgent reperfusion can save viable myocardial tissue and reduce morbidity and mortality. ECGs should be performed within 10 minutes of clinical contact and be interpreted by experienced clinicians, with remote processes in place if needed. Continuous cardiac monitoring is recommended for people at high risk and should be regularly reviewed. For those with non-ischaemic ECGs, resolved symptoms and normal troponin levels, monitoring is not required. Repeated 12-lead ECGs should be done at intervals or if symptoms change [54–56]. Evidence supporting the recommendations
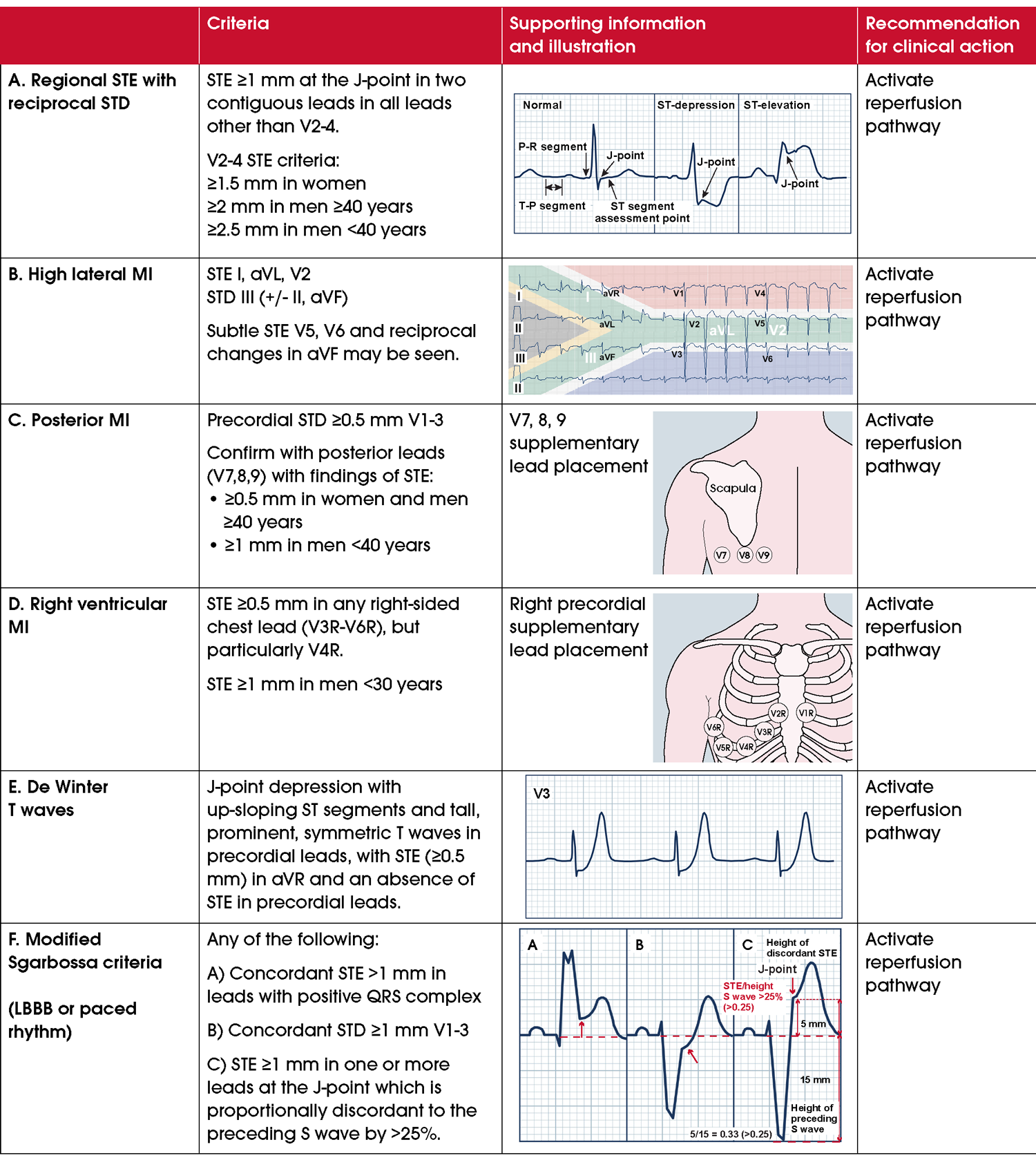
ECG findings of acute coronary occlusion myocardial infarction
ST-segment elevation (STE) is the key ECG criterion required to determine whether reperfusion is warranted (Figure 4A) [57]. STE is not specific to ACOMI and may occur in other cardiac and non-cardiac disease states [58]. These include pericarditis, left ventricular hypertrophy (LVH), left ventricular aneurysm, left bundle branch block (LBBB), right ventricular pacing, Takotsubo or other cardiomyopathies and Brugada patterns. Non-cardiac STE conditions include normal variant STE (early repolarisation), pulmonary embolism, hyperkalaemia, hypothermia and raised intracranial pressure.In the clinical context of myocardial ischaemia, STE should be assumed to represent ACOMI until proven otherwise.
In addition to STE criteria, there are other ECG patterns indicative of an ACOMI [59–61]. Recognising these improves accurate ECG detection rates for acute coronary occlusion and may prompt consideration for reperfusion (Figure 4) [62]. Supplementary lead ECGs may be needed to interrogate areas of the heart such as the inferior, basal, posterior and right ventricular walls (Table 4). Additional ECG information and findings are included in the Comprehensive Guideline.Detecting new STE with an abnormal baseline ECG
ECG evidence for ACOMI may be difficult to discern in people with LBBB, right ventricular pacing or LVH.
The validated Modified Sgarbossa criteria improves diagnosis of STE in people with LBBB or right ventricular pacing. The criteria can discern STE with a specificity of 99% and a sensitivity of 80% in these populations (Figure 4F) [63–65]. The Modified Sgarbossa criteria has a straightforward threshold, STE exceeding >25% of the depth of the preceding S wave in any lead.Currently, there are no validated methods to distinguish STE due to ACOMI, LVH or hypertrophic cardiomyopathy using an ECG alone. Clinical suspicion for ACOMI should be high in the presence of haemodynamic compromise and/or symptoms consistent with ACS.
In people with LVH, current and historical ECGs should be compared. If historical ECG data are unavailable, continuous cardiac monitoring, close clinical observation and repeated ECGs are required to monitor for development of acute coronary occlusion. Expert consultation should be sought for people with persisting ischaemic symptoms and equivocal ECG findings of ACOMI.
Figure 4 ECG findings consistent with acute coronary occlusion myocardial infarction (ACOMI). Abbreviations: ECG, electrocardiogram; LBBB, left bundle branch block; MI, myocardial infarction; STD, ST-segment depression; STE, ST-segment elevation.
| Cardiac region | Leads with STE | Reciprocal STD |
| Anterolateral | I, aVL, V3–V6 | II, III, aVF |
| Anteroseptal | V1–V4 | None |
| Septal | V1, V2 | None |
| Inferior | II, III, aVF | I, aVL |
| Right ventricular | Right-sided chest leads V3–6 | |
| Posterior | Posterior leads V7–9 | V1–V3 |
| Lateral | I, aVL, V5, V6 | II, III, aVF |
| High lateral | I, aVL (V2) | III (II, aVF) |
Abbreviations: ECG, electrocardiogram; STE, ST-segment elevation; STD, ST-segment depression.
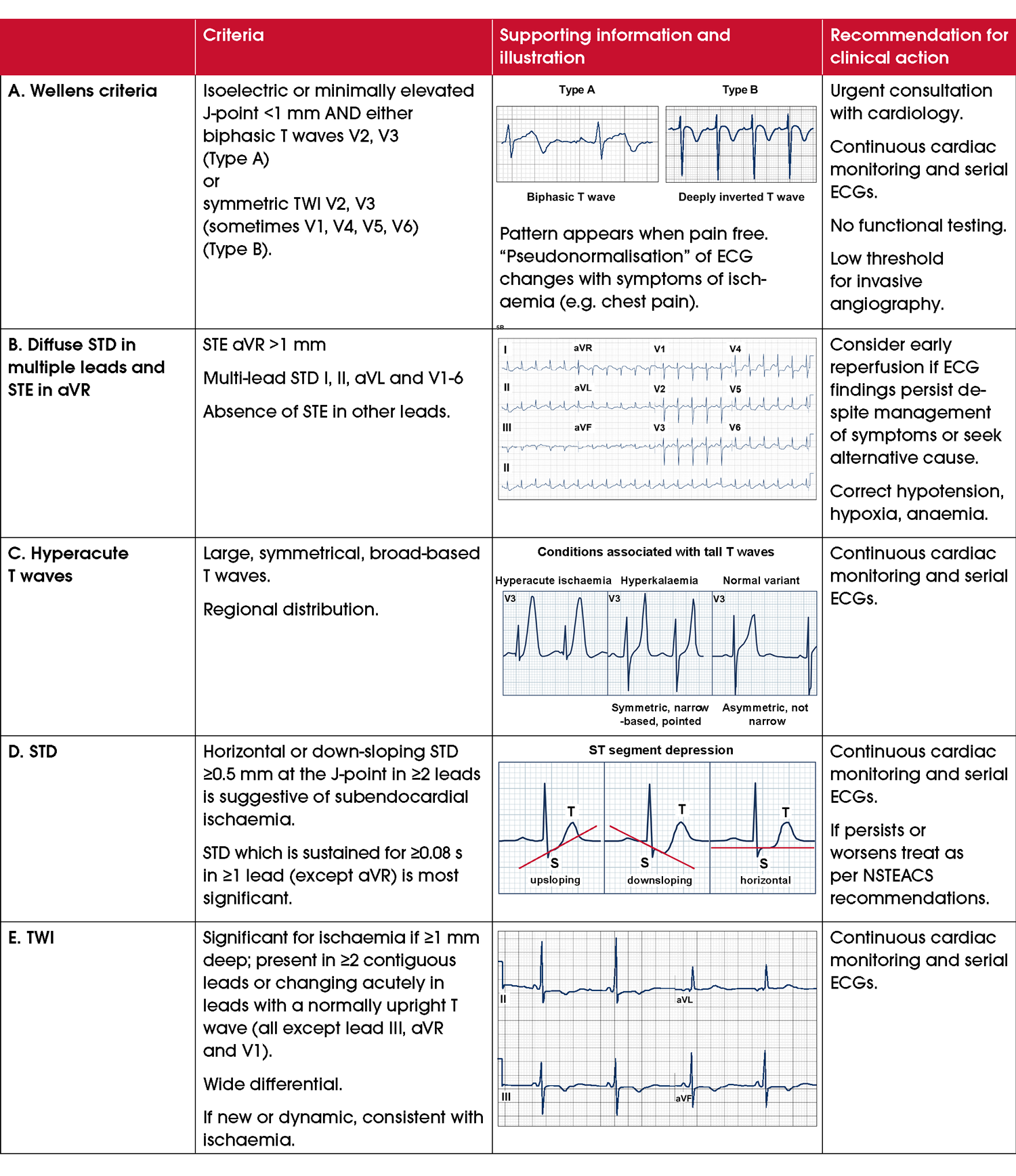
High-risk ECG findings
Acute coronary occlusion may not be evident on the initial ECG. Certain ECG patterns are associated with potential progression to ACOMI. They require prompt and continuous clinical ECG monitoring.
Wellens T waves: defined by characteristic T wave inversions in the precordial leads. Where symptoms have resolved, these inversions may indicate a reperfusion syndrome linked to severe stenosis of the left anterior descending artery, known as Wellens syndrome (Figure 5A) [66]. In such cases, avoid provocative tests (for example, exercise stress testing) and consider invasive coronary angiography. If or when ischaemic symptoms recur, the ECG recorded during those symptoms will often appear pseudonormalised, with T waves becoming more upright. Diffuse ST-segment depression across multiple leads with STE in aVR: may represent global ischaemia of various etiologies including a left main occlusion, triple vessel disease or oxygen supply/demand mismatch ischaemia seen in type 2 MI (Figure 5B) [67]. People with persisting symptoms with no identifiable alternative causes of ischaemia or who do not respond to treatment of alternative causes (e.g. hypoxia, anaemia, hypotension) should be considered for coronary angiography [68]. Hyperacute T waves: symmetrical, broad-based T waves disproportionately large to the preceding QRS complex can be the first ECG finding of an evolving MI, although its prognostic significance has been questioned (Figure 5C) [69, 70]. These people should be subject to close clinical and continuous cardiac monitoring and serial 12-lead ECGs to examine for signs of ACOMI. An important differential diagnosis is hyperkalaemia (Figure 5C).Other signs of myocardial ischaemia on ECG
Additional ECG findings in a person with suspected myocardial ischaemia which warrant continuous cardiac monitoring and consideration of treatment for NSTEACS include:
- ST-segment depression: ≥0.5 mm at the J-point in ≥2 contiguous leads which is horizontal or downsloping (Figure 5D). The deeper and more widespread the depression, the more severe the ischaemia [71, 72]. ST-segment depression in contiguous leads should be first considered as reciprocal change of ACOMI and the ECG examined for corresponding STE, as ST-segment depression secondary to subendocardial ischaemia does not generally localise to a regional coronary territory (Figure 4, Figure 5 and Table 4) [73]. Although ST-segment depression occurs in other conditions (e.g. LVH, hypokalaemia, digoxin use), a systematic review found it to be highly specific (97.2–99.3%) but poorly sensitive (16.6–20.0%) for ischaemia [74].
- T wave abnormalities: including dynamic inversion or flattening (Figure 5E). New T wave inversion compared to a previous ECG or dynamic T wave changes during serial ECGs may represent ischaemia. Specificity of T wave inversion for ischaemia is higher in the context of other signs of ischaemia on the ECG [75].
Figure 5 High-risk ECG findings for ACS and findings suggestive of cardiac ischaemia. Abbreviations: ECG, electrocardiogram; NSTEACS, non-ST-segment elevation acute coronary syndromes; STD, ST-segment depression; STE, ST-segment elevation; TWI, T wave inversion.
Computer-assisted ECG interpretation
There is currently no international standardised system for computer-based ECG interpretation. Manufacturers of ECG machines use distinct algorithms, leading to variability in sensitivity and specificity for diagnosing cardiac conditions. Common computer-assisted interpretation errors in diagnosing ACS and ACOMI include misattributing Q waves associated with LVH, LBBB and/or dilated or hypertrophic cardiomyopathy to ACOMI. Additionally, errors often fail to distinguish between STE caused by early repolarisation, pericarditis, or LBBB and ACOMI [76]. Accurate ECG interpretation by a clinician, with consideration of the clinical context, remains essential.Continuous ECG monitoring
Continuous ECG monitoring is not required in people with no ongoing symptoms, normal or non-ischaemic ECG changes and initial normal troponin values. In people with suspected ACS, ongoing ECG monitoring is recommended for those at high-risk (see Table 5).
| Haemodynamic instability or cardiogenic shock |
| Recurrent or ongoing chest pain refractory to medical treatment |
| Cardiac arrest |
| Recurrent dynamic ST-T wave changes or other changes consistent with myocardial ischaemia or infarction on ECG |
| Arrhythmias (e.g. sustained ventricular tachycardia, high-degree atrioventricular block) |
| Mechanical complications of MI (e.g. new systolic murmur) |
| Acute heart failure |
Abbreviations: ACS, acute coronary syndromes; AMI, acute myocardial infarction.
Practice points
- Do not rely on an initial normal ECG to exclude ACS. If myocardial ischaemia is strongly suspected, record and interpret serial ECGs.
- Do not delay treatment decisions/treatment in people with symptoms of ischaemia and clear evidence of ACOMI on ECG while waiting for troponin test results.
- Do ensure consistent lead placement when performing serial ECGs to prevent artefactual errors in interpretation.
Future direction
Artificial intelligence (AI) and machine learning has been applied to ECG and clinical data with the aim of delivering a more accurate and timely assessment of ACS and ACOMI [77]. Its utility remains in research at this time.Biomarkers
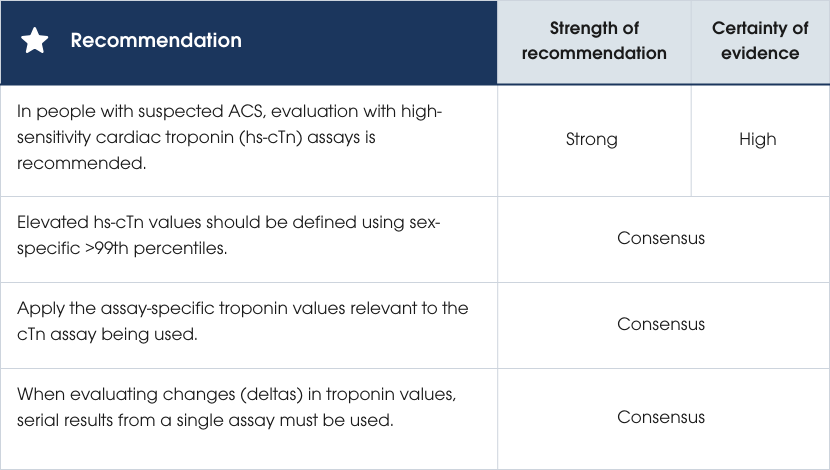
Recommendations
Evidence supporting the recommendations
High-sensitivity cardiac troponin (hs-cTnI or hs-cTnT) is the preferred biomarker for diagnosing ACS due to its precision, early detection of myocardial injury and improved accuracy for MI. These assays enable faster decision-making, reduce unnecessary admissions and account for sex differences (see Section Risk assessment and clinical decision pathways for suspected ACS). If unavailable, contemporary troponin assays can be used with longer testing intervals and clinical risk assessment [1, 78–90].Evidence supporting the recommendations
Analytic properties of cardiac troponin assays
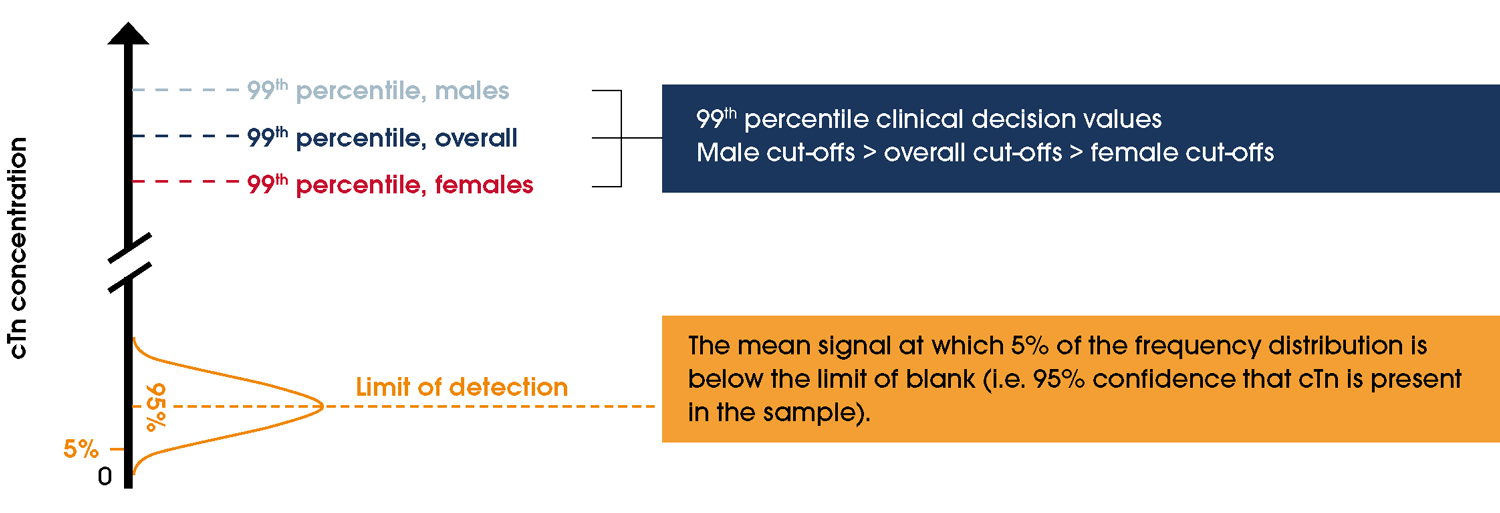
The cut-off or threshold indicative of myocardial injury is a cTn value above the assay-specific 99th percentile derived from a healthy population [1]. Contemporary assays in Australia use µg/L, while high-sensitivity assays use ng/L, reflecting their greater sensitivity. Other performance metrics, such as the limit of detection and limit of blank, are also critical for evaluating assay reliability (see Table 6, Figure 6). Women have lower circulating cTn concentrations, resulting in sex-specific 99th percentile values [91]. Using older, non-sex-specific cut-off values can lead to underdiagnosis of myocardial injury and MI in women [92].| Characteristic | Contemporary troponin assays* | High-sensitivity troponin assays |
| Precision | Variable | ≤10% CV at 99th percentile |
| Detection | ~20–50% of healthy reference population | ≥50% of healthy reference population |
| Units | Microgram per litre (µg/L) | Nanogram per litre (ng/L) |
| Sex-specific 99th percentiles | No. Overall 99th percentile values only | Yes. Female and male 99th percentiles |
| Timing of serial testing for MI using 99th percentile | 0 and 6–8 hours | 0 and 3 hours |
| Single low-risk troponin values for MI* | No | Yes |
| Ability to use in rapid, early assessment strategies | No | Yes |
| Platform | POC and laboratory-based | POC and laboratory-based |
*Using a contemporary assay, if a person presents symptom-free for >6–8 hours, only one test is needed. If ≤99th percentile at 6–8 hours, no second test is required. If >99th, a second test is needed.
Abbreviations: cTn, cardiac troponin; CV, coefficient of variation; MI, myocardial infarction; POC, point-of-care.
Point-of-care troponin assays
Contemporary POC troponin assays require serial measurements over 6–8 hours in people with suspected ACS. POC troponin assays may lead to more timely management of people with suspected ACS, with comparable safety to laboratory-based assays (Table 7) [88–90, 97]. Early data support rapid assessment using POC hs-cTn assays [88–90, 98]. Knowledge of POC hs-cTn assays is rapidly evolving. Their use in clinical decision pathways in EDs, outpatient clinics and primary care may become more common given the clinical safety and cost efficacy of such assays [97].Figure 6 Various analytic definitions for troponin assays. Note: Adapted with permission from Januzzi et al. [99]. Abbreviations: cTn, cardiac troponin.Clinical interpretation of troponin values
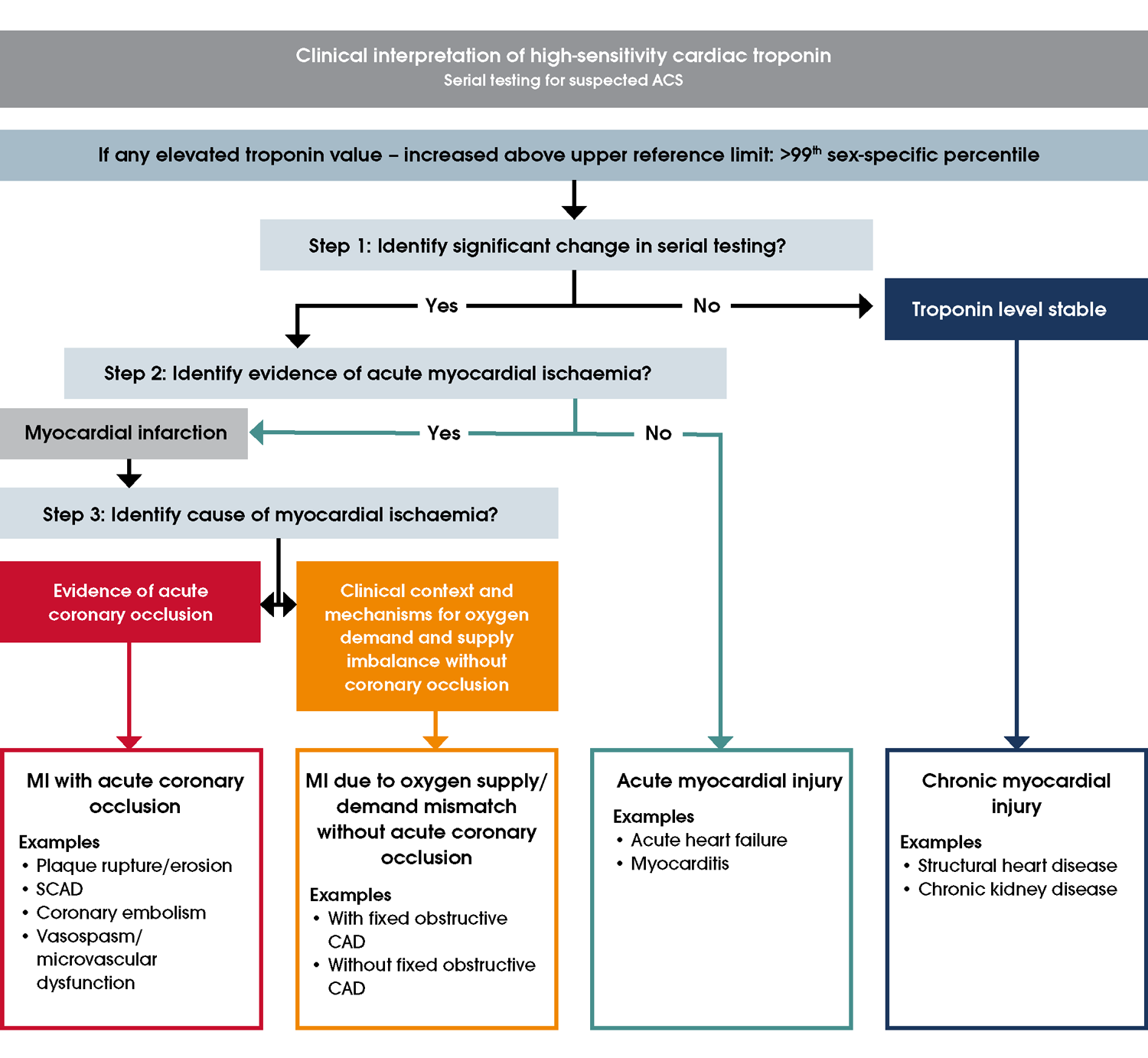
cTn results must be interpreted alongside the clinical context and ECG findings (Figure 7) [27]. Serial measurements are required to track whether cTn elevation is stable or changing. Stable elevation occurs with chronic myocardial injury as well as in the plateau phase of troponin release in MI (such as when presentation was delayed). People with changing values (increasing or decreasing) warrant evaluation for evidence of myocardial ischaemia. Acute myocardial injury due to other causes (e.g. acute heart failure, pulmonary embolism) needs to be considered. Differentiation between MI subtypes and other myocardial injury requires careful evaluation (Figure 7) [100]. The introduction of hs-cTn assays has led to a decrease in the proportion of people with UA, defined with cTn values ≤99th percentile, and many who would previously be classified as UA are now found to have MI [101].Figure 7 Clinical interpretation of high-sensitivity cardiac troponin (hs-cTn) results. Adapted with permission from the Accelerated Chest Pain Risk Evaluation (ACRE) Project, Clinical Excellence Queensland, Queensland Health. For guidance on identifying evidence for acute myocardial ischaemia, refer to sections High-risk ECG findings and Other signs of myocardial ischaemia on ECG. Abbreviations: ACS, acute coronary syndromes; CAD, coronary artery disease; MI, myocardial infarction; SCAD, spontaneous coronary artery dissection.
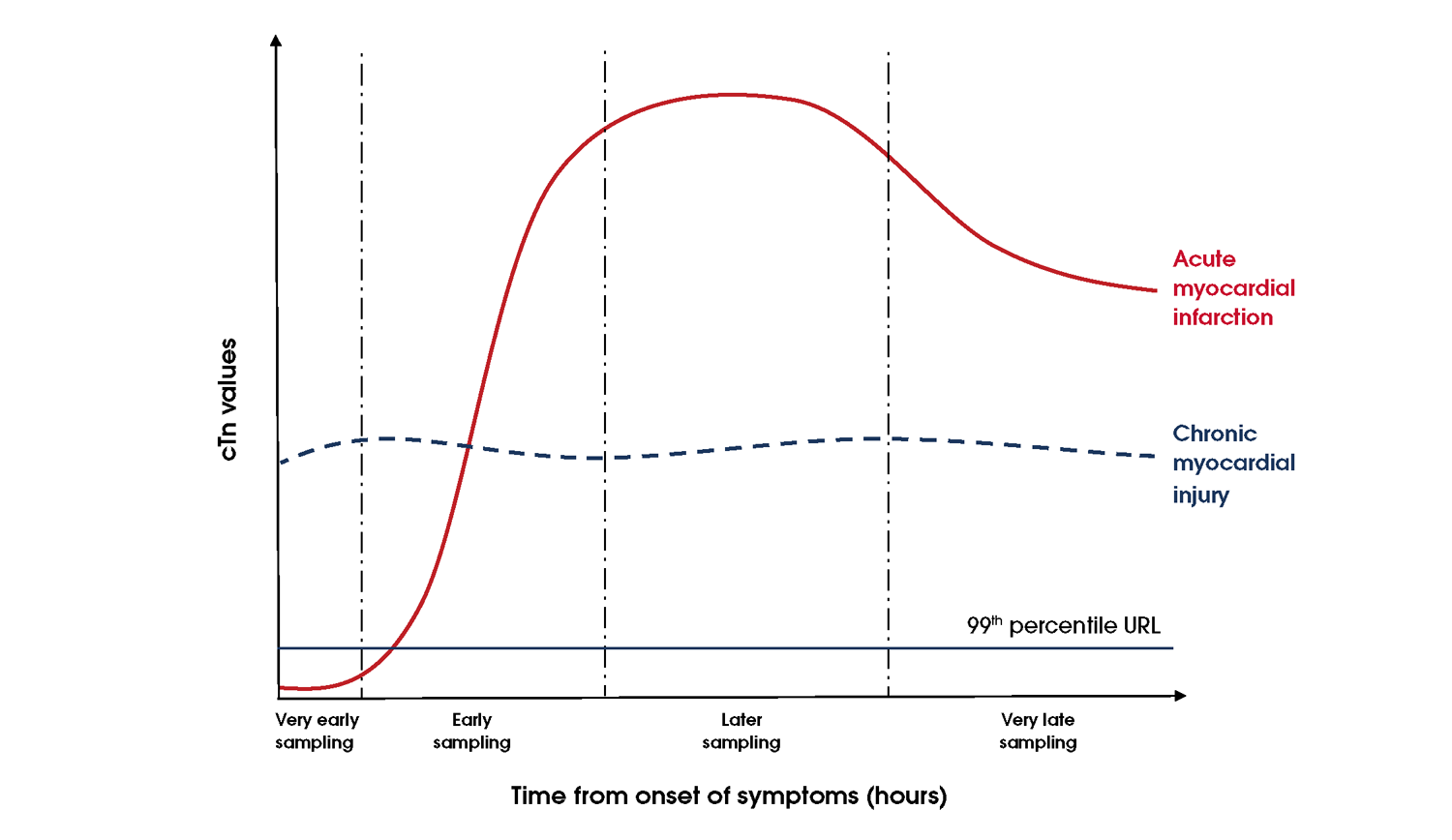
Time from onset of coronary occlusion vs symptom onset
In the setting of ACOMI, there may be a delay in elevation of cTn levels. This delay has shortened with more sensitive tests, including hs-cTn assays, able to detect elevations earlier (Figure 8). Repeat troponin testing is required for people with ongoing or recurrent symptoms or where there is a high suspicion of ACS.
Figure 8 Early troponin kinetics in people with acute myocardial infarction. Abbreviations: cTn, cardiac troponin; URL, upper reference limit.
Comparing results from different troponin assays
cTn assays developed by various diagnostic companies use different antibody combinations, resulting in different numerical results for the same amount of circulating troponin [102]. Results of one assay cannot be interpreted using the reference range of a different assay. Serial testing of cTn concentrations can only be interpreted when measured using the same assay.Differences between troponin T and I assays
High-sensitivity cardiac troponin I (hs-cTnI) and high-sensitivity cardiac troponin T (hs-cTnT) have comparable accuracy for the early detection and diagnoses of MI [103]. Troponin T is more likely to be elevated among people with poor renal function (see Renal disease) and chronic muscular diseases (e.g. chronic myopathy, myositis). This is possibly due to re-expression of cTnT in the diseased muscle or due to cross reactivity of the cTnT assay with skeletal muscle troponin T [104, 105].Non-MI causes of troponin elevation
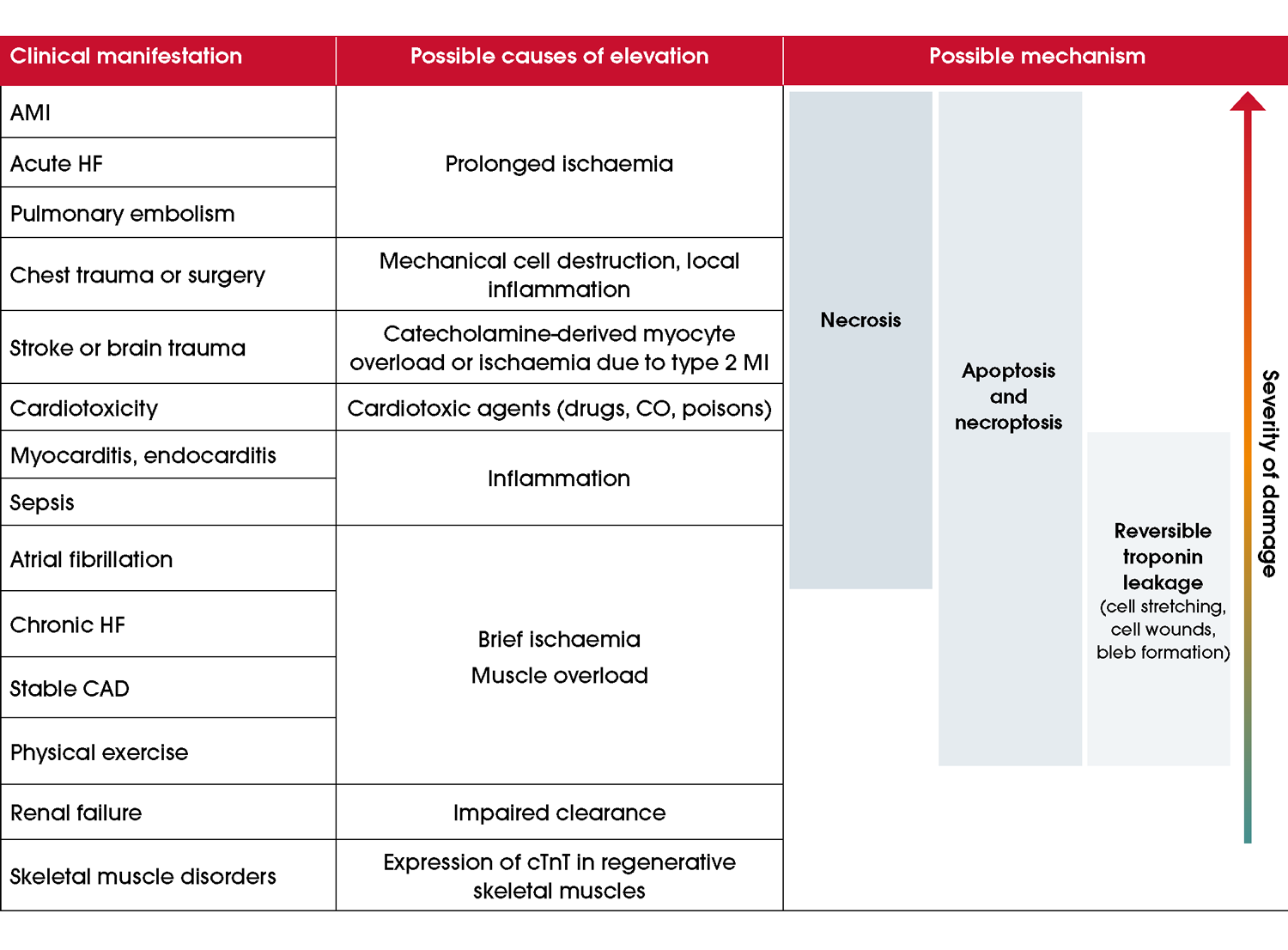
Numerous ischaemic, non-coronary cardiac and non-cardiac causes of myocardial injury can result in elevated cTn concentrations (Figure 9) [1, 80, 106, 107]. Life-threatening conditions including aortic dissection and pulmonary embolism may result in elevated cTn values. Cardiac troponin elevation indicates myocardial injury but is not specific to the underlying pathophysiology [1]. Figure 9 Conditions associated with troponin elevation. Adapted from Katrukha et al. [69]. Abbreviations: AMI, acute myocardial infarction; CAD, coronary artery disease; CO, carbon monoxide; cTnT, cardiac troponin T; HF, heart failure; MI, myocardial infarction.Renal disease
Chronically elevated cTn concentrations are often reported with decreased renal function, more so cTnT than cTnI. MI diagnosis in people undergoing haemodialysis requires serial cTn measurements, rather than management according to an elevated baseline value [108].False positive and false negative cardiac troponin results
False positive or negative cTn results are rare but possible. False positives may occur due to antibody interference, such as macrotroponins – high molecular weight complexes of cTn fragments and immunoglobulins (cTn autoantibodies) – which delay troponin clearance and cause artificially elevated readings. Heterophilic antibodies cause another type of interference, as these can bind to test antibodies and yield a positive result without actual cTn elevation. While the exact cause of heterophilic antibodies is unclear, they are sometimes associated with conditions like rheumatoid arthritis or viral infections, including Epstein–Barr virus and cytomegalovirus [109]. Conversely, severe haemolysis or plasma substances like biotin can lead to false negatives. If troponin levels do not align with the clinical presentation, consulting the hospital laboratory is essential to rule out these rare false positive cTn results [110, 111].Other biomarkers
Additional biomarkers exist but are not used to diagnose MI. There is no role for creatine kinase MB-isoenzyme (CK-MB) to identify reinfarction in people with AMI [112].Risk assessment and clinical decision pathways for suspected ACS
Recommendations
Recommendations
Evidence supporting the recommendations
Clinical decision pathways improve care and efficiency in suspected ACS by identifying MI and those at high-risk of MACE within 30 days (e.g. those requiring further investigation) while reducing unnecessary tests and admissions for low-risk individuals. Structured risk assessments incorporating clinical data, troponin and ECG findings achieve missed MI or 30-day MACE rates of <1% [23, 113]. People with suspected ACS or ECGs suggestive of ischaemia, or high-risk features, should undergo inpatient evaluation [23, 114–116]. Clinical decision pathways for people without ACOMI or ischaemic ECG findings include those based on hs-cTn results alone or clinical risk scores like the Emergency Department Assessment of Chest Pain Score (EDACS) and history, ECG, age, risk factors and troponin (HEART) [117]. Evidence supporting the recommendations
Clinical decision pathways
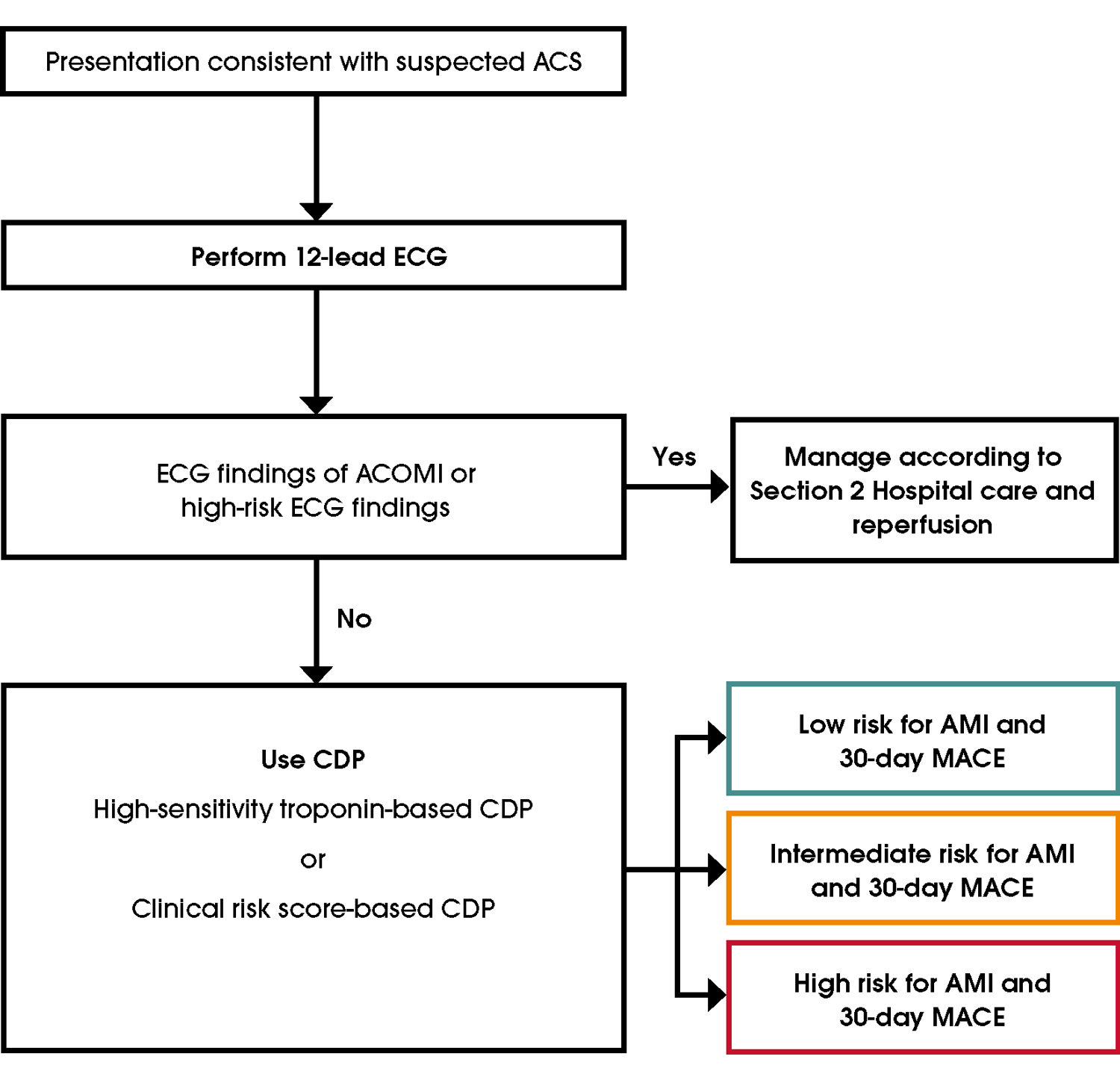
Risk stratification
A three-tiered stratification system groups people into high, intermediate or low risk of MACE, including MI (Figure 10). This system uses cTn levels, clinical history, physical examination and ECG findings (normal, non-ischaemic or unchanged from previously).
High risk: Risk of a 30-day event, most commonly MI, exceeds 50–70% [118]. Admission and further evaluation are required. Not all high-risk individuals have MI, so clear communication about risk is essential. Refer to Section 2 Hospital care and reperfusion for further information on the evaluation and management of high-risk people. Intermediate risk: These people have a 30-day MACE risk of 2–22% using hs-cTn-based decision pathways (0/1 or 0/2-hour strategies) and require further evaluation [119–121]. Serial cTn values of ≤99th percentile allow outpatient testing, as the 30-day MACE rate is <2% (Section Further diagnostic testing for people with suspected ACS) [23]. Elevated (>99th percentile) cTn values require evaluation in an inpatient setting. Elevated but stable cTn values consistent with chronic myocardial injury increases long-term cardiac risk without MI, and therefore is beyond this guideline's scope (Section Biomarkers) [1, 122]. Low risk: The 30-day MACE risk is <1% using hs-cTn-based clinical decision pathway or clinical risk score (Supplementary Material B2). hs-cTn strategies identify more low-risk individuals than contemporary cTn pathways. In low-risk people defined by a hs-cTn strategy, further testing to exclude AMI is not required [27, 123, 124].Figure 10 Assessment process for people with suspected ACS. For further information, refer to Initial ECG assessment, High-sensitivity troponin-based clinical decision pathways and Clinical score-based clinical decision pathways in the guideline. Abbreviations: ACOMI, acute coronary occlusion myocardial infarction; ACS, acute coronary syndromes; AMI, acute myocardial infarction; CDP, clinical decision pathway; ECG, electrocardiogram; MACE, major adverse cardiovascular events.
Risk stratification for people with suspected ACS: identifying MI and UA
For people without findings consistent with ACOMI on the initial ECG, further assessment aims to identify NSTEMI and UA through evaluation of clinical features, additional ECGs and troponin testing. NSTEMI is associated with elevated cTn values.
People with ongoing or recurrent ischaemic symptoms, or new ECG findings suggestive of ischaemia during initial or repeat testing, should be classified as high risk for ACS. If clinical suspicion remains high, serial cTn testing is recommended, as late cTn rises have been described in <1% of people with NSTEMI [125].High-sensitivity troponin-based clinical decision pathways
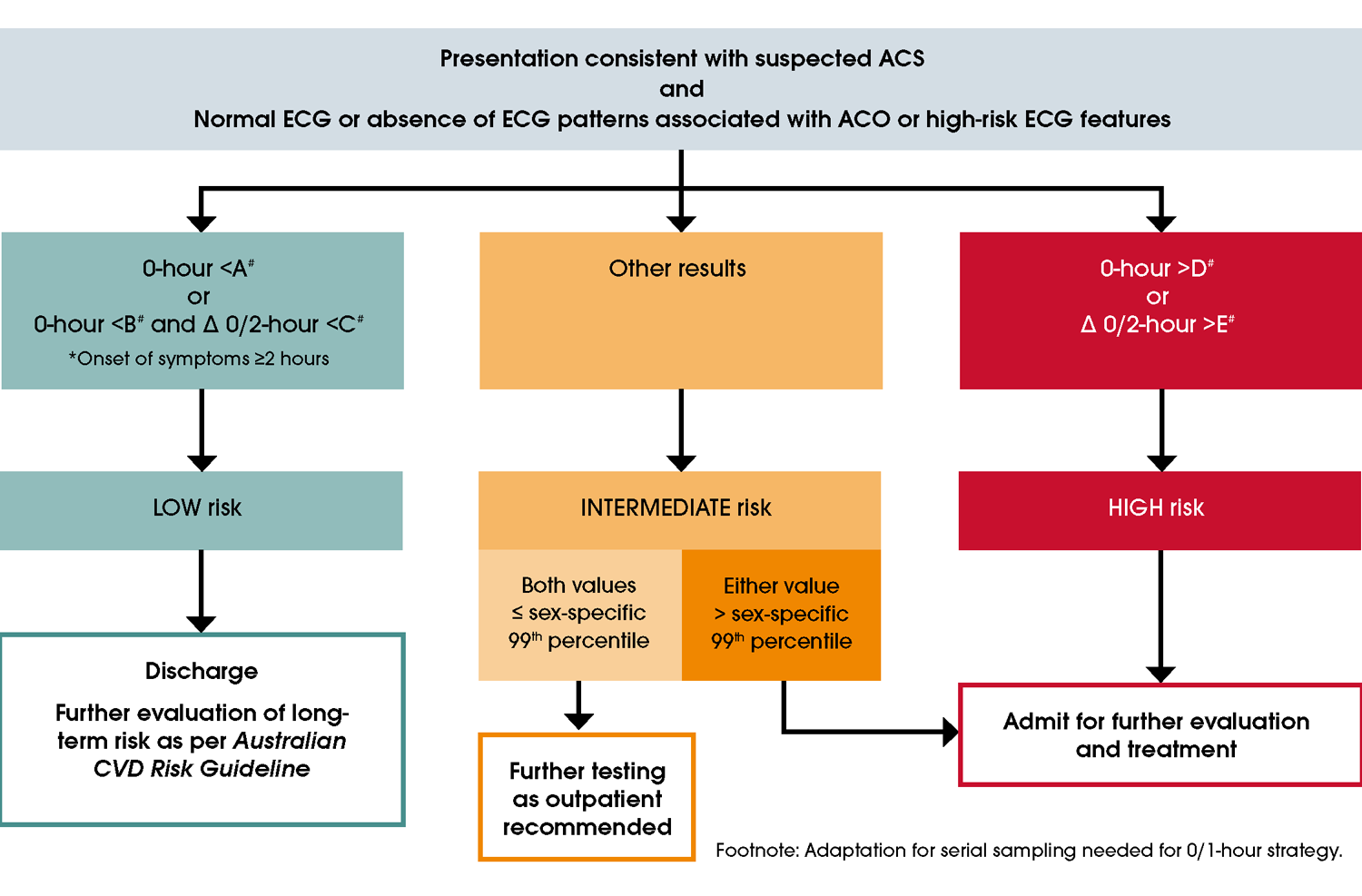
The use of hs-cTn assays is recommended over contemporary troponin assays for safe and rapid decision-making. In people presenting with chest pain, hs-cTn-based risk stratification typically identifies 50–65% as low risk, 20–30% as intermediate risk and 15–25% as high risk for MACE [126, 127]. When combined with non-ischaemic ECG findings in validated algorithms, stratification of an individual’s risk of adverse cardiac event can safely and effectively be achieved without clinical risk scores [27, 118]. The 0-hour, 0/1-hour and 0/2-hour protocols are time-critical pathways designed to reduce myocardial damage and improve outcomes, using hs-cTn levels and clinical history to guide decisions. These strategies have been developed for most hs-cTn assays and the values are assay-specific, with details provided in Figure 11, Table 7 and Supplementary Material B1 [26, 83, 118, 120, 125, 128–135].Single high-sensitivity cardiac troponin measurements
A single hs-cTn measurement is not suitable to guide treatment decisions for people with symptom onset <2 hours. These people require serial testing [120].[83, 132, 136–139]. In people with symptom onset ≥2 hours, combining a single hs-cTn result with non-ischaemic ECG findings can very safely classify 20–50% of people presenting with possible ACS as low risk [26, 31, 78, 82, 120, 124, 126–129, 132, 139–146]. Single hs-cTnT and hs-cTnI assays have been extensively validated, demonstrating high negative predictive value and sensitivity for excluding index MI and a <1% risk of MACE during short- and longer-term follow-up [24, 81, 82, 124, 126, 139, 146–149]. Unlike hs-cTn assays, single contemporary troponin measurements have not been validated to assess risk [150].0/1- and 0/2-hour strategies
Index or 30-day MACE rates range between 2–22% for people identified as intermediate risk using the 0/1- or 0/2-hour strategies. Those deemed intermediate risk require additional evaluation (refer to Section Further diagnostic testing for people with suspected ACS) [26, 120, 121]. For those with normal serial cTn values, 30-day MACE rates are ≤2% [23]. While primarily evaluated in large observational studies, randomised trials of the 0/1-hour strategy have demonstrated 30-day MI and death rates of <1% when implemented successfully [26, 131, 151–153]. The thresholds for changes (deltas) in the 0/1- and 0/2-hour algorithms are both assay and time dependent, making collection of blood specimens within the specified windows critical (Table 7).In most hospitals, delays in central laboratory assay turnaround times render the 0/1-hour strategy impractical. POC hs-cTn assays may overcome this limitation but are not yet widely available. A 0/2-hour strategy is therefore currently the most practical option in most settings.
Figure 11 hs-cTn 0/2-hour testing recommendations. Note: the 0/2-hour time points are shown in this figure. If using a 0/1-hour strategy, change timeframes accordingly. #Refer to Table 7 interpretation of cTn assay-specific values and sex-specific 99th percentiles. *All people with symptom onset <2 hours need serial testing. People with ongoing symptoms should be assessed according to high-risk criteria. Abbreviations: ACO, acute coronary occlusion; ACS, acute coronary syndromes; ECG, electrocardiogram.
| Assay | Sampling timepoints | A | B | C | D | E | Female 99th percentile (F) | Male 99th percentile (G) |
|---|---|---|---|---|---|---|---|---|
| Hs-cTnI (Architect; Abbott) | 0/1h | <4 | <5 | <2 | ≥64 | ≥6 | 16 | 34 |
| 0/2h | <4 | <6 | <2 | ≥64 | ≥15 | 16 | 34 | |
| Hs-cTnI (Access; Beckman Coulter) | 0/1h | <4 | <5 | <4 | ≥50 | ≥15 | 11 | 20 |
| 0/2h | <4 | <5 | <5 | ≥50 | ≥20 | 11 | 20 | |
| Hs-cTnI (Centaur; Siemens) | 0/1h | <3 | <6 | <3 | ≥120 | ≥12 | 40 | 58 |
| 0/2h | <3 | <8 | <7 | ≥120 | ≥20 | 40 | 58 | |
| Hs-cTnI (Atellica; Siemens) | 0/1h | <4 | <6 | <3 | ≥120 | ≥12 | 39 | 54 |
| 0/2h | NA | NA | NA | NA | NA | NA | NA | |
| Hs-cTnI (Vitros; Clinical Diagnostics) | 0/1h | <1 | <2 | <1 | ≥40 | ≥4 | 9 | 12 |
| 0/2h | <1 | <2 | <3 | ≥40 | ≥5 | 9 | 12 | |
| Hs-cTnT (Elecsys; Roche) | 0/1h | <5 | <12 | <3 | ≥52 | ≥5 | 9 | 17 |
| 0/2h | <5 | <14 | <4 | ≥52 | ≥10 | 9 | 17 | |
| Hs-cTnI (Pathfast; LSI Medience)* | 0/1h | <3 | <4 | <3 | ≥90 | ≥20 | 20 | 30 |
| 0/2h | <3 | TBD | TBD | ≥90 | TBD | 20 | 30 | |
| Hs-cTnI (Triage True; Quidel)* | 0/1h | <4 | <5 | <3 | ≥60 | ≥8 | 14 | 26 |
| 0/2h | <4 | TBD | TBD | ≥60 | TBD | 14 | 26 | |
| Hs-cTnI (VTLi, Siemens)* | 0/1h | <4 | TBD | TBD | TBD | TBD | 18 | 27 |
| 0/2h | <4 | <6 | <5 | ≥60 | ≥15 | 18 | 27 |
Abbreviations: hs-cTnI, high-sensitivity cardiac troponin I; hs-cTnT, high-sensitivity cardiac troponin T; NA, not available; TBD, to be determined.
High-STEACS algorithm
The United Kingdom (UK) High-STEACS algorithm is a validated, safe and effective approach for the diagnosis and management of ACS using a variety of hs-cTn assays [24, 124, 127, 131, 138]. Further details are described in the Supplementary Material B1.Clinical score-based clinical decision pathways
Sites using contemporary cTn assays
Clinical score-based tools, such as EDACS and the HEART score, are recommended for assessing people with suspected ACS when using contemporary cTn assays. These tools are the most widely validated, demonstrating high sensitivity for index AMI and 30-day MACE (see Supplementary Material B2) [142, 144, 161–169]. Compared to hs-cTn-based clinical decision pathways, clinical score-based tools identify fewer low- or intermediate-risk individuals and may require additional testing without significantly improving MACE outcomes [31, 78, 119, 128, 129, 140, 145, 150, 160, 170, 171]. Furthermore, because sex-specific considerations are not included in all scoring systems, their effectiveness in men and women may not be equal [172]. Further information on these clinical score-based tools is provided in the Supplementary Material B2.Implementing a clinical decision pathway for suspected ACS
Multidisciplinary teams and clinical decision pathways for suspected ACS, especially those using hs-cTn assays, offer substantial benefits for people and healthcare systems [23, 123, 135, 137, 153, 164, 165, 173–176]. For example:- Australian randomised control trial data showed that when using hs-cTnT, the 0/1-hour strategy resulted in more frequent ED discharge and a reduced ED length of stay. Similar clinical outcomes occurred at 30 days compared to usual care (0/3-hour cTn measurements with an hs-cTnT threshold of ≥30 ng/L) [151].
- A large, randomised trial showed failure to follow recommended management processes for low-risk people increased resource use with no differences in 30-day MACE [123].
Centres choosing to implement an alternate strategy to the recommended CDPs should validate the chosen clinical decision pathways. Validation requires evaluating 30-day mortality and re-presentation with confirmed ACS in all people who presented with chest pain.
Practice points
Women
- Apply sex-specific 99th percentile upper reference limits when using hs-cTn assays [1, 24, 92, 119, 133, 177].
- Women are frequently misdiagnosed with non-ischaemic chest pain, and their ACS risk is often underestimated [25, 30, 172, 178, 179]. This is due in part to traditional risk tools lacking sex-specific considerations and clinician bias.
Older adults
- Use uniform hs-cTn cut-offs for clinical assessment, recognising that concentrations increase with age in healthy individuals. This may result in fewer older adults being classified as low risk for MI [1].
- In people aged 65 years and older with comorbidities such as renal impairment, the specificity of hs-cTn assays for MI is reduced [26, 130, 177, 180].
First Nations peoples
- Use a single hs-cTn measurement in First Nations peoples to identify people as low risk of MI and 30-day MACE [93].
- Be cautious when implementing the HEART score and IMPACT pathways, which have been evaluated in small studies in First Nations populations (see Supplementary Material B2) [181].
- Investigate all First Nations adults (aged 18 years and over) with suspected ACS for underlying CAD, given their high risk of future cardiac events [182, 183].
People with renal impairment
- Use hs-cTn-based strategies in people with renal dysfunction, noting that fewer individuals will be classified as low risk compared to other approaches [26, 180].
- Elevations in cTn are common in this population, leading to their exclusion from many assessment trials [184]. The safety of hs-cTn-based strategies appears to be similar in people with and without renal dysfunction.
Future direction
Newer strategies for individualised determination of likelihood of MI
Newer strategies to determine risk of MI have been developed based on large international datasets using machine learning techniques. These include the MI3 algorithm study, the ARTEMIS study and the CoDE-ACS study [185–187]. Such strategies incorporate additional information (e.g. specific interval time of cTn testing, biometric measurements) to support decisions. Validation studies suggest large proportions of people can be defined as low risk, with improved specificity for MI in high-risk people.Initial therapeutic management
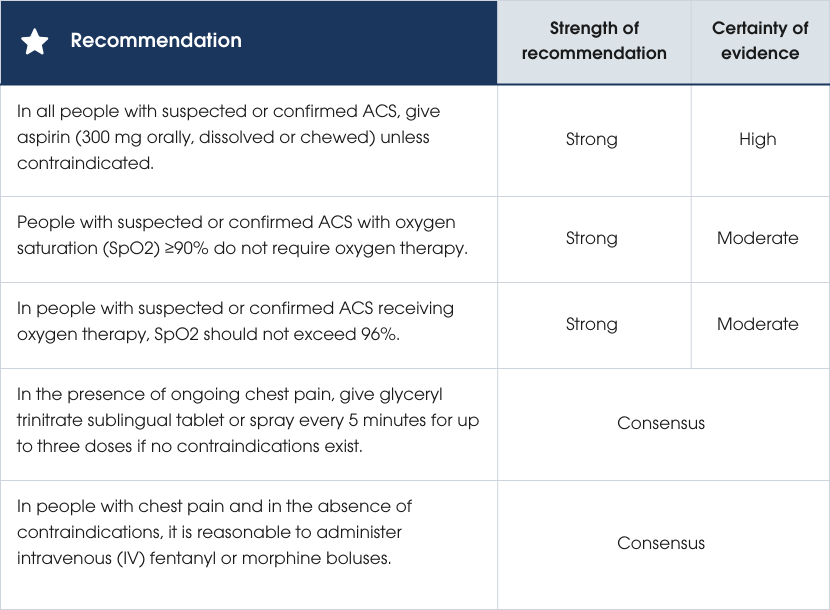
Recommendations
Recommendations
Evidence supporting the recommendations
Evidence supporting the recommendations
Aspirin
Aspirin reduces the risk of vascular events (vascular death, MI and stroke) in individuals with ASCVD, with benefits outweighing the small risk of major bleeding. A 300 mg loading dose is recommended to fully inhibit platelet activation, followed by a maintenance dose of 100 mg, which is as effective as higher doses [188–190].Oxygen therapy
Routine supplemental oxygen for suspected ACS without hypoxaemia does not improve mortality at 30 days or 12 months and is associated with increased risk of recurrent MI and revascularisation. Higher oxygen saturation levels are cautioned due to a dose–response link with increased mortality in acute and intensive care settings [191–193].Practice points
Oxygen therapy
- Provide oxygen therapy routinely if oxygen saturation falls below 90%, as hypoxaemia at this level is assumed to contribute to coronary ischaemia. The clinical outcome benefits remain uncertain [191].
- Recognise that oxygen therapy is commonly administered when oxygen saturation is 90–92%, although its benefit is unknown [194–196].
- Exercise caution in people with chronic obstructive pulmonary disease (COPD) when administering supplemental oxygen, aiming for a target arterial oxygen saturation (SaO2) of approximately 88–92%.
Nitrates
- Use IV glyceryl trinitrate for more effective symptom relief in acute ischaemia compared to the sublingual form but be aware that it does not improve prognosis [197, 198].
- Do not administer glyceryl trinitrate in cases of hypotension, right ventricular infarction or recent use of phosphodiesterase 5 inhibitors (e.g. sildenafil, vardenafil, tadalafil). Consider alternative therapy if symptoms persist.
Opioid analgesia
- Titrate opioid doses to resolve chest pain, adjusting for individual needs based on age, comorbidities and concurrent medication use [199–201].
- Consider fentanyl for its short time to peak effect, short duration of action and minimal impact on histamine release. Note that both morphine and fentanyl are associated with increased platelet reactivity, reduced antiplatelet effect of P2Y12 inhibitors and slower absorption of oral medicines such as ticagrelor during the early hours of ACS [200, 202–204].
Other medicines
- Do not administer non-steroidal anti-inflammatory drugs (NSAIDs) in confirmed ACS, especially during the early phase, due to an increased risk of MACE [205, 206].
- Do not initiate additional antiplatelet, anticoagulation or beta blocker therapies without a confirmed or probable diagnosis of ACS. For further information refer to Section Antiplatelet therapy in the acute phase and Anticoagulant therapy in the acute phase.
Further diagnostic testing for people with suspected ACS
Recommendations
Recommendations
Evidence supporting the recommendations
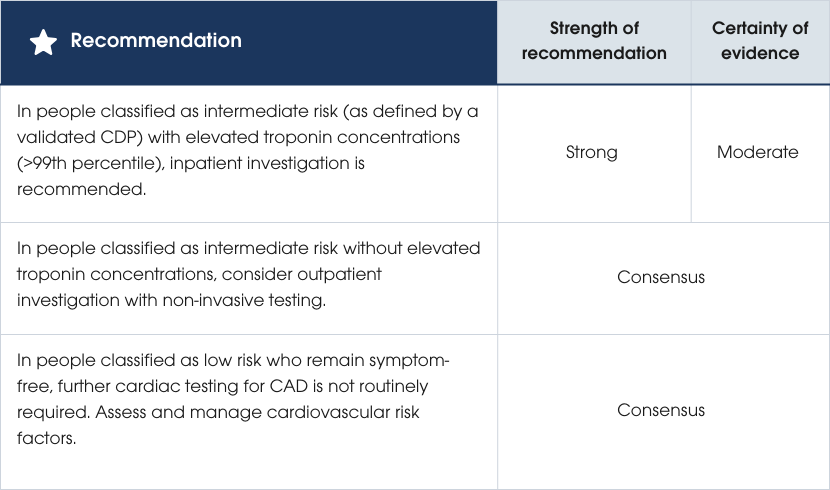
For people at intermediate risk, invasive angiography or non-invasive cardiac testing is recommended to refine risk stratification, identify alternative causes of chest pain and assess future risk of ACS beyond 30 days. Inpatients with elevated hs-cTn levels above the 99th percentile should undergo testing due to a 30-day cardiac event rate of 2–22%, while those with hs-cTn ≤99th percentile may consider outpatient testing within 30 days, as their event rate is <2% [16]. Non-invasive testing is not routinely recommended for low-risk individuals, as their likelihood of cardiac events over two years is minimal. General practitioner follow-up is advised for symptom resolution, treatment and assessment of long-term cardiovascular risk using Australian guidelines (cvdcheck.org.au) [23, 27, 89, 119, 123, 124, 126, 138, 147, 207–211]. Evidence supporting the recommendations
Practice points
Non-invasive test selection – anatomical versus functional
Anatomical testing (CTCA)
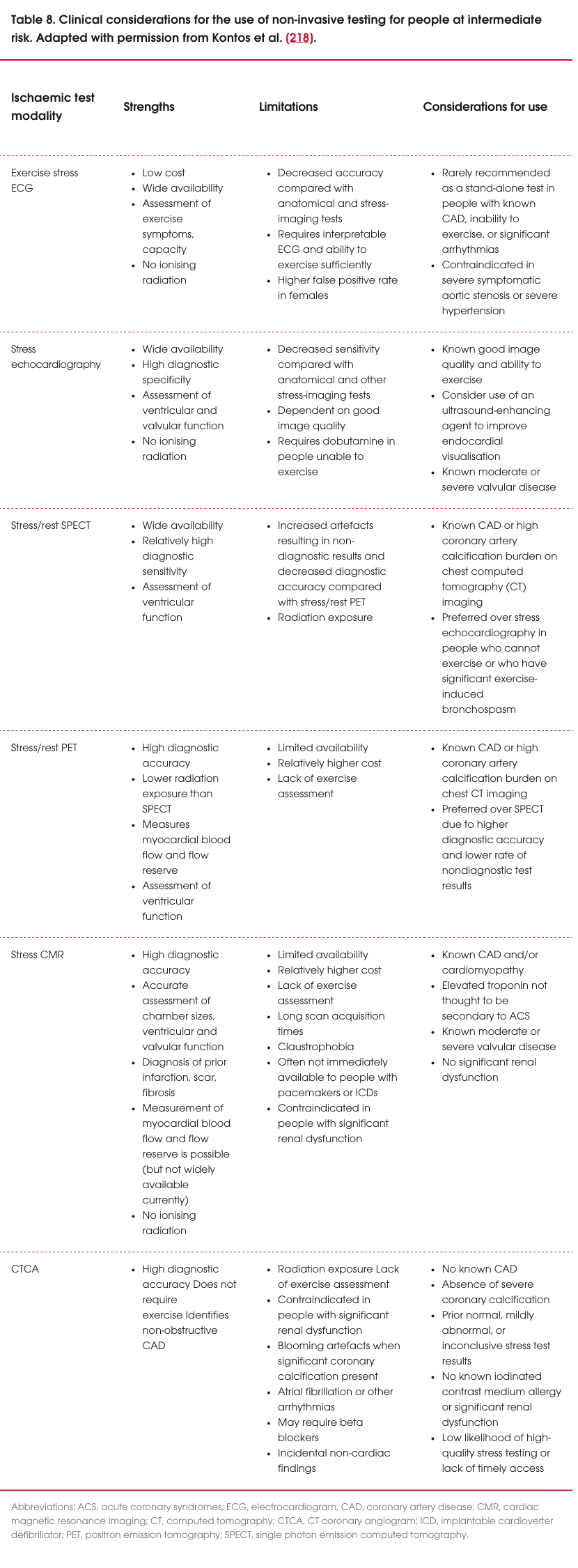
- Use computed tomography coronary angiography (CTCA) as a first-line investigation for people without previously known coronary artery disease (CAD) presenting with intermediate-risk ACS, if no contraindications exist (see Table 8).
- Consider functional testing with close monitoring and a graduated exercise regime for people with contraindications to CTCA.
- Recognise that a normal CTCA (ruling out both obstructive and non-obstructive plaque) reliably excludes ACS and indicates an extremely low risk of ACS for at least 4–5 years [212–217]. Identifying non-obstructive plaque on CTCA can guide preventative therapies, such as lipid-lowering treatment.
- Do not rely on coronary artery calcium scoring alone in ACS evaluation unless combined with CTCA.
Functional testing
- Favour functional testing for people with known CAD, prior stents or extensive coronary calcification, where CTCA interpretation may be more challenging. Functional tests can help identify whether symptoms are caused by obstructive plaque and assess ischaemic burden and short-term prognosis.
- Select functional tests based on clinical needs, including stress echocardiography, stress cardiac magnetic resonance imaging (MRI), stress/rest single-photon emission computed tomography (SPECT), stress/rest positron emission tomography (PET), or exercise ECG (see Table 8).
- Recognise the additional diagnostic benefits of stress cardiac MRI and echocardiography, such as evaluating left ventricular function, regional wall motion abnormalities and valvular function, and excluding differential diagnoses like myopericarditis and Takotsubo cardiomyopathy.
Considerations for test selection
- Consider individual cardiovascular risk factors, local expertise and the availability of health services, particularly in regional and remote areas, when selecting non-invasive cardiac investigations as these all may influence the selection [27, 218–220].
- Prioritise inpatient non-invasive testing for low-risk individuals with factors limiting access to timely follow-up or the ability to re-present to ED, such as First Nations peoples with suspected ACS or those facing sociodemographic challenges [182].
Cost-effectiveness
Reducing unnecessary testing has benefits for the individual and health services. For example, an Australian study estimated a total cost saving of $13.5 million per annum after implementation of an accelerated diagnostic pathway (using cTnl, ECG and TIMI score) that reduced hospital admission rates and ED length of stay [165].Considerations for regional, remote and First Nations peoples
Regional, remote and First Nations peoples are disproportionately affected by reduced access to healthcare services, longer wait times and greater travel distances to diagnostic services. Definitive early identification of CAD using CTCA may be of significant benefit in this group because it is a relatively more accessible imaging technology [221]. To note, there is limited evidence on how long a negative CTCA ensures low risk before retesting. An Australian telemedicine program has demonstrated the potential to reduce waiting times by supporting remote exercise stress testing with specialist cardiology support. This initiative has enabled a significant number of people to be managed within their local health facilities, improving access and reducing the need for travel [222].Role of rapid access chest pain clinics
Rapid access chest pain clinics provide screening, investigations and management for people presenting with chest pain, including those discharged after an ACS [222]. Studies from the UK report these clinics as safe, efficient and cost-effective alternatives to hospital admission [223]. Australian models of chest pain clinics have shown comparable outcomes, demonstrating similar benefits regardless of referral patterns or specific investigations undertaken [224]. These services offer improved access to diagnostic tests, individual satisfaction, and cost savings. They have shown safety outcomes comparable to, or better than, traditional hospital-based care, with reduced rates of invasive investigations, fewer ED re-presentations, and streamlined follow-up of test results [225–229]. Access should be prioritised for selected people at intermediate risk with cTn levels below the 99th percentile.Re-presentation with symptoms
People re-presenting to ED within 30 days with possible ACS symptoms without prior non-invasive testing for CAD and/or coronary ischaemia may warrant further functional or anatomical testing. A detailed reassessment for alternate diagnoses is also required. If prior exercise ECG testing was negative, more sensitive and specific investigations or anatomical tests should be considered.
Primary care and regional and remote presentations
Recommendations
Recommendations
Evidence supporting the recommendations
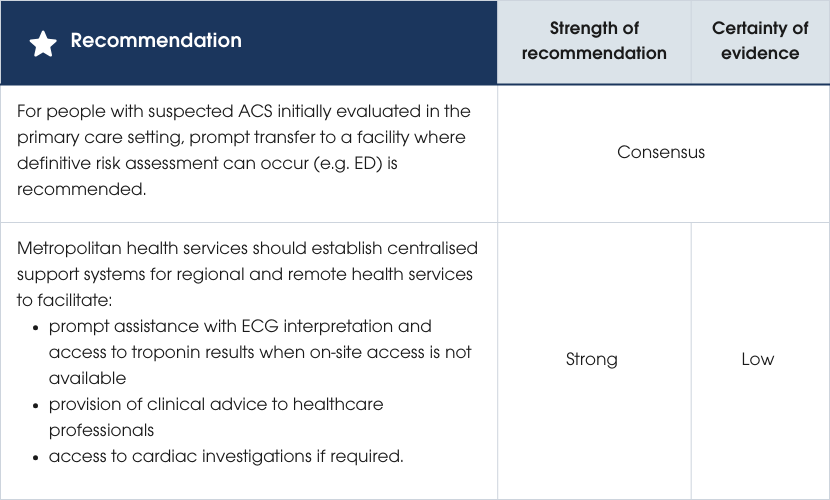
All individuals with suspected ACS should have access to best-practice care, regardless of location. Centralised and coordinated care systems, supported by telehealth, ensure prompt specialist input for services outside tertiary centres [230]. An Australian model demonstrated reduced mortality with early cardiologist support for ECG interpretation, POC troponin testing and decision-making. Data also show fewer missed STEMIs when tertiary-level support was routinely available compared to usual care in hospitals without an emergency physician [55]. Evidence supporting the recommendations
Considerations for primary care presentation
Initial assessment
As outlined previously, assessment of ACS incorporates:
- ECG findings
- clinical findings from history and physical examination
- troponin testing.
The ability of a healthcare professional or clinic to reliably diagnose or exclude ACS is determined by their capacity to perform and interpret these components. If the clinician suspects ACS, transfer to the nearest medical facility where capacity for definitive assessment for ACS can occur is mandatory.
Initial ECG assessment
People presenting with suspected ACS require prompt access to an ECG (within 10 minutes) and interpretation by a suitably trained clinician. If ACS is suspected, recording an ECG should not delay transfer to a facility that can perform serial troponin testing and provide reperfusion therapy, as delays are associated with greater harm [231–236].If an ECG cannot be performed within 10 minutes, prompt transfer via ambulance to a location where an ECG can be performed is necessary. This may mean the first ECG is evaluated by trained paramedics.
If an ECG is non-ischaemic and the clinical presentation does not align with ACS as the likely diagnosis, it is reasonable to continue assessment in the primary care setting.
Troponin testing
If ACS remains a possible diagnosis after initial history, examination and ECG assessment, and cTn testing is required, the person should be transferred to the nearest facility (usually an ED) for definitive risk assessment [22, 27, 237]. In Australia, there are no single test strategies using POC contemporary troponin assays to exclude AMI [238] Serial testing is required and typically cannot be performed in the primary care setting. hs-cTn POC assays are available, but not widely distributed. Currently, there is limited evidence for single test strategies to exclude MI in primary care settings [90].Risk assessment and clinical decision pathways
Risk scores such as the Marburg Heart Score, Grijseels and Bruins Slot rules are not recommended for excluding ACS in a primary care setting [239]. A systematic review of older risk assessment rules without cTn results found no difference between the use of these scores and a general practitioner's clinical judgement in ruling out ACS [240].Considerations for regional and remote presentations
While the initial assessment remains unchanged, key factors influencing the decision to transfer people with suspected ACS include [20, 21, 241, 242]:- local service capabilities and support availability in regional and remote settings
- availability of relevant investigations including chest X-rays, cTn testing and/or other cardiac tests (e.g. CTCA, exercise stress testing, echocardiography)
- the healthcare professional’s clinical judgement.
Initial ECG assessment
If the ECG can be performed but not interpreted, it is reasonable to seek urgent remote evaluation (e.g. via telehealth) [55].When the clinical and ECG assessment supports a diagnosis of ACOMI, consideration of urgent reperfusion therapy is required. Urgent transfer of the person to the nearest facility for fibrinolysis or primary PCI is needed (refer to Section 2 Hospital care and reperfusion).
Troponin testing
Many regional and remote settings are reliant on contemporary cTn assays, including POC platforms [113]. Clinicians must be aware of the type of troponin assay in use locally and ensure results are used in an evidence-based clinical decision pathway (refer to Section Biomarkers and Section Risk assessment and clinical decision pathways for suspected ACS). Where contemporary cTn assays are in use, UA should be considered in the presence of normal cTn results if clinical suspicion for ACS is high based on ECG interpretation and/or clinical history. Further serial cTn testing over 6–8 hours should occur [22]. Management may include initial treatment for presumed ACS, a period of continuous cardiac monitoring and/or transfer to a PCI-capable centre.Risk assessment and clinical decision pathways
Evidence supporting the use of clinical risk scores without incorporating troponin values is limited. In the absence of hs-cTn assays, incorporation of contemporary cTn results with clinical risk scores within a validated clinical decision pathway is crucial (refer to Section Clinical score-based clinical decision pathways and Supplementary Material B2). In a rural New Zealand setting, use of the EDACS accelerated diagnostic pathway with serial POC contemporary cTn measurements safely stratified risk in people with suspected ACS (see Supplementary Material B2) [243].Further diagnostic testing
If a particular diagnostic test is required but unavailable regionally, transfer to another facility should be considered.
Tertiary centres have an obligation to support appropriate testing in people from regional and remote areas. Decisions on further diagnostic testing can be informed by consultation with metropolitan cardiac teams or, when available, rapid access chest pain clinics. Remote access to such clinics may help improve diagnostic pathways for people in regional and remote settings.
Discharge planning and advice
Following a comprehensive and structured assessment, people with suspected ACS who do not require admission for further assessment and/or management can be discharged.
The outcome of ED assessment will determine the guidance provided to the person (and their support people) prior to discharge.
While many people will not receive a definitive diagnosis for their symptoms, life-threatening conditions like AMI and UA will have been deemed to be of very low probability [244]. Specific discharge advice for non-ACS presentations is beyond the scope of this guideline Discharge planning and advice supports reduced ED presentations and leads to better outcomes. Clinicians, including registered nurses and nurse practitioners, should be supported to undertake comprehensive pre-discharge assessment and discharge planning. This is important to help manage a person’s anxiety; high levels of anxiety are associated with an increased likelihood of symptom recurrence and re-presentation to hospital [245, 246].Discharge communication for general practitioners
To support ongoing management of people after discharge, concise information in the form of a discharge summary must be promptly provided to a person’s general practitioner.
Discharge advice for people at low risk for ACS
During discharge, people classified as low risk should be provided with written information and verbal advice that includes:
- evidence-based information on the condition(s) diagnosed and any further investigation/management required
- clear communication that the person has been comprehensively assessed to exclude AMI and to determine UA is unlikely
- a clear statement that CAD has not been excluded, and that follow-up with the person’s general practitioner is recommended for assessment and management according to the Australian guideline for assessing and managing cardiovascular disease risk (cvdcheck.org.au)
- information about what steps the person should take if they experience recurrent symptoms
- education on cardiovascular health and cardiovascular causes of chest pain and other key symptoms
- guidance on where to find reliable sources of online health information and how to contact telephone-based triage services.
Discharge advice for people at intermediate risk for ACS
During discharge, people classified as intermediate risk should be provided with written information and verbal advice that includes:
- clear reassurance for the person and their support people that they have been comprehensively assessed as safe to be discharged
- clear information on management of existing and/or new symptoms, including when to call an ambulance, re-present to the ED or contact their general practitioner
- information on referral for outpatient assessment and management; this should include clear guidance on the clinician/clinic referred to, and whether an appointment has already been arranged or whether the person or support people need to do this; if the latter, then a clear timeline and contact details to arrange the appointment should be provided in the written discharge advice
- clear guidance on new or continuing medicines, including when and how to take them
- education on cardiovascular disease risk and steps that the person can take to reduce their risk
- guidance on where to find reliable sources of online health information and how to contact telephone-based triage services.
Discharge advice for people with a prior history of coronary heart disease who have a chronic or stable coronary syndrome
A small number of people presenting to the ED with a prior diagnosis of CAD may be discharged with probable chronic or stable CAD. The advice given needs to balance:
- reassurance that the person has been comprehensively assessed and is deemed safe for discharge at this time, notwithstanding a probable cardiac cause for their symptoms and presentation
- a clear plan for further follow-up for assessment and management
- a clear plan for managing existing or new symptoms
- education on their cardiovascular health and reiteration of when to call an ambulance, re-present to ED or contact their general practitioner.
Preamble
2. Hospital care and reperfusion